Magnesium »
PDB 101d-1an0 »
1a95 »
Magnesium in PDB 1a95: Xprtase From E. Coli Complexed with Mg:Cprpp and Guanine
Enzymatic activity of Xprtase From E. Coli Complexed with Mg:Cprpp and Guanine
All present enzymatic activity of Xprtase From E. Coli Complexed with Mg:Cprpp and Guanine:
2.4.2.22;
2.4.2.22;
Protein crystallography data
The structure of Xprtase From E. Coli Complexed with Mg:Cprpp and Guanine, PDB code: 1a95
was solved by
S.Vos,
R.J.Parry,
M.R.Burns,
J.De Jersey,
J.L.Martin,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 50.00 / 2.00 |
| Space group | P 43 21 2 |
| Cell size a, b, c (Å), α, β, γ (°) | 94.200, 94.200, 165.900, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 20.7 / 23.2 |
Magnesium Binding Sites:
The binding sites of Magnesium atom in the Xprtase From E. Coli Complexed with Mg:Cprpp and Guanine
(pdb code 1a95). This binding sites where shown within
5.0 Angstroms radius around Magnesium atom.
In total 2 binding sites of Magnesium where determined in the Xprtase From E. Coli Complexed with Mg:Cprpp and Guanine, PDB code: 1a95:
Jump to Magnesium binding site number: 1; 2;
In total 2 binding sites of Magnesium where determined in the Xprtase From E. Coli Complexed with Mg:Cprpp and Guanine, PDB code: 1a95:
Jump to Magnesium binding site number: 1; 2;
Magnesium binding site 1 out of 2 in 1a95
Go back to
Magnesium binding site 1 out
of 2 in the Xprtase From E. Coli Complexed with Mg:Cprpp and Guanine
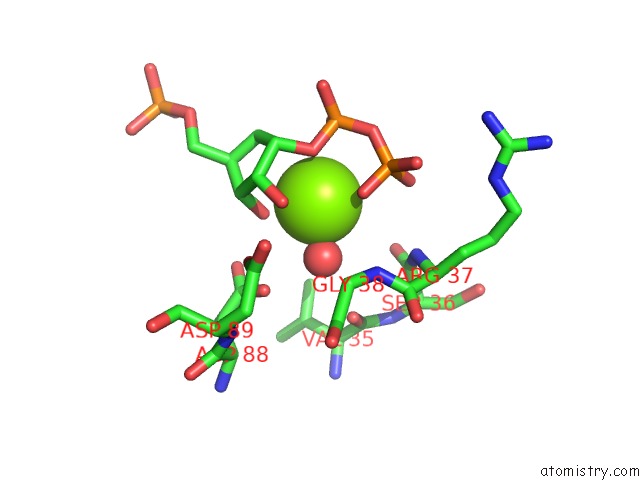
Mono view
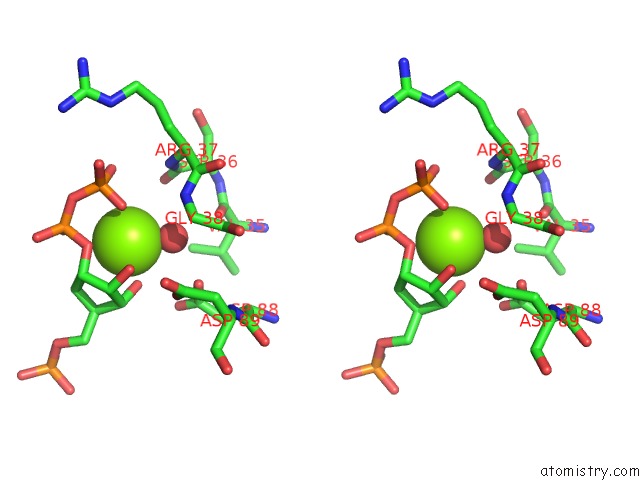
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 1 of Xprtase From E. Coli Complexed with Mg:Cprpp and Guanine within 5.0Å range:
|
Magnesium binding site 2 out of 2 in 1a95
Go back to
Magnesium binding site 2 out
of 2 in the Xprtase From E. Coli Complexed with Mg:Cprpp and Guanine
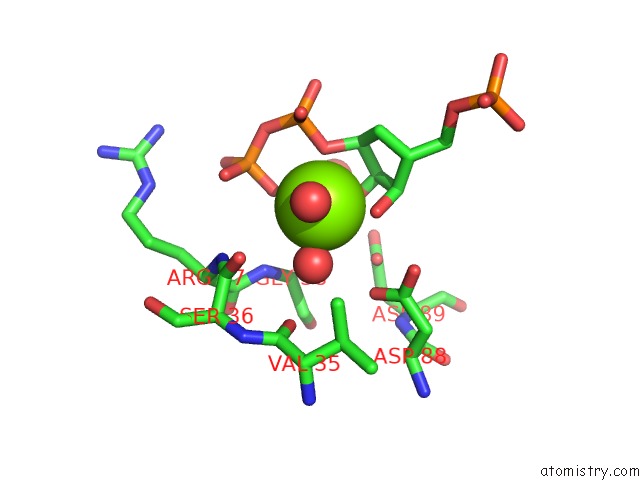
Mono view
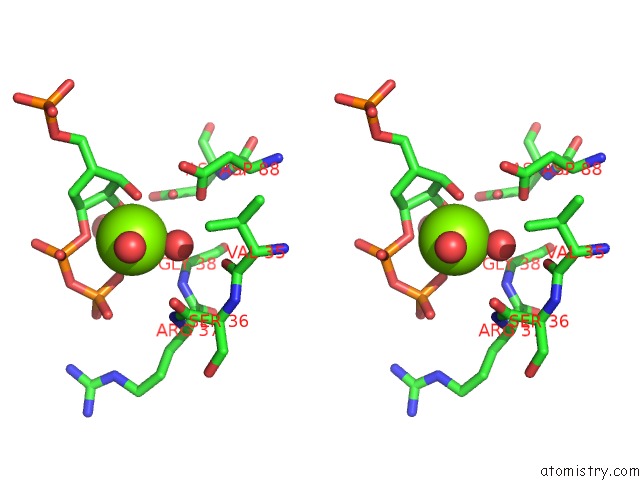
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 2 of Xprtase From E. Coli Complexed with Mg:Cprpp and Guanine within 5.0Å range:
|
Reference:
S.Vos,
R.J.Parry,
M.R.Burns,
J.De Jersey,
J.L.Martin.
Structures of Free and Complexed Forms of Escherichia Coli Xanthine-Guanine Phosphoribosyltransferase. J.Mol.Biol. V. 282 875 1998.
ISSN: ISSN 0022-2836
PubMed: 9743633
DOI: 10.1006/JMBI.1998.2051
Page generated: Sat Aug 9 20:03:25 2025
ISSN: ISSN 0022-2836
PubMed: 9743633
DOI: 10.1006/JMBI.1998.2051
Last articles
Mg in 3CCUMg in 3CCS
Mg in 3CCR
Mg in 3CCQ
Mg in 3CCM
Mg in 3CCL
Mg in 3CCJ
Mg in 3CC2
Mg in 3CCE
Mg in 3CC7