Magnesium »
PDB 1gq9-1h7q »
1gro »
Magnesium in PDB 1gro: Regulatory and Catalytic Mechanisms in Escherichia Coli Isocitrate Dehydrogenase: Multiple Roles For N115
Enzymatic activity of Regulatory and Catalytic Mechanisms in Escherichia Coli Isocitrate Dehydrogenase: Multiple Roles For N115
All present enzymatic activity of Regulatory and Catalytic Mechanisms in Escherichia Coli Isocitrate Dehydrogenase: Multiple Roles For N115:
1.1.1.42;
1.1.1.42;
Protein crystallography data
The structure of Regulatory and Catalytic Mechanisms in Escherichia Coli Isocitrate Dehydrogenase: Multiple Roles For N115, PDB code: 1gro
was solved by
J.A.Grobler,
J.H.Hurley,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 6.00 / 2.50 |
| Space group | P 43 21 2 |
| Cell size a, b, c (Å), α, β, γ (°) | 105.100, 105.100, 150.600, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 19.5 / n/a |
Magnesium Binding Sites:
The binding sites of Magnesium atom in the Regulatory and Catalytic Mechanisms in Escherichia Coli Isocitrate Dehydrogenase: Multiple Roles For N115
(pdb code 1gro). This binding sites where shown within
5.0 Angstroms radius around Magnesium atom.
In total only one binding site of Magnesium was determined in the Regulatory and Catalytic Mechanisms in Escherichia Coli Isocitrate Dehydrogenase: Multiple Roles For N115, PDB code: 1gro:
In total only one binding site of Magnesium was determined in the Regulatory and Catalytic Mechanisms in Escherichia Coli Isocitrate Dehydrogenase: Multiple Roles For N115, PDB code: 1gro:
Magnesium binding site 1 out of 1 in 1gro
Go back to
Magnesium binding site 1 out
of 1 in the Regulatory and Catalytic Mechanisms in Escherichia Coli Isocitrate Dehydrogenase: Multiple Roles For N115
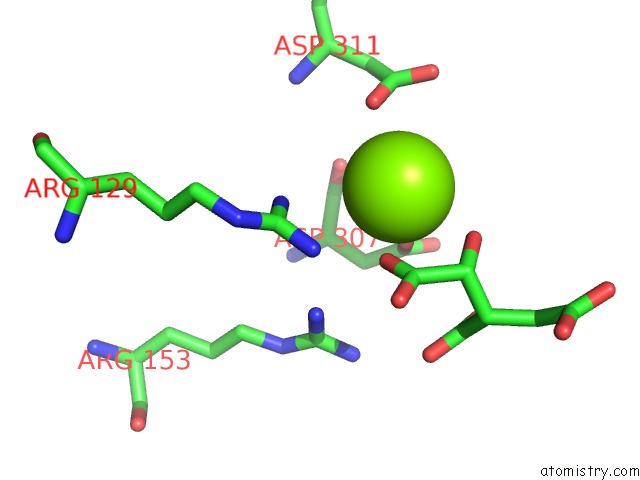
Mono view
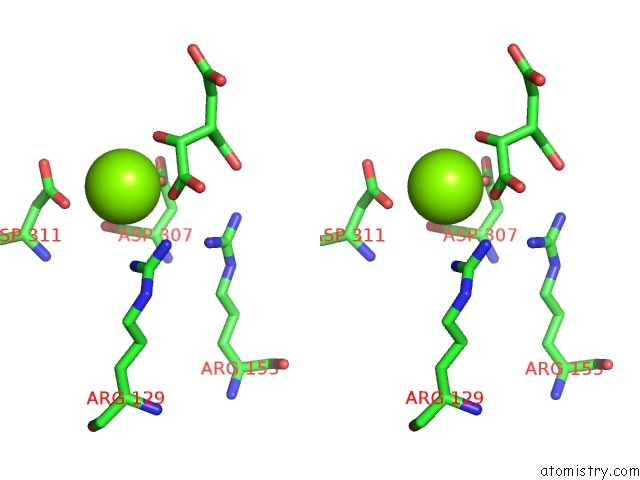
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 1 of Regulatory and Catalytic Mechanisms in Escherichia Coli Isocitrate Dehydrogenase: Multiple Roles For N115 within 5.0Å range:
|
Reference:
R.Chen,
J.A.Grobler,
J.H.Hurley,
A.M.Dean.
Second-Site Suppression of Regulatory Phosphorylation in Escherichia Coli Isocitrate Dehydrogenase. Protein Sci. V. 5 287 1996.
ISSN: ISSN 0961-8368
PubMed: 8745407
Page generated: Tue Aug 13 03:49:55 2024
ISSN: ISSN 0961-8368
PubMed: 8745407
Last articles
F in 4LY6F in 4MM8
F in 4MIH
F in 4MM4
F in 4MK0
F in 4MIG
F in 4MH0
F in 4MDD
F in 4MDB
F in 4MC9