Magnesium »
PDB 2f6y-2fl2 »
2fhy »
Magnesium in PDB 2fhy: Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor
Enzymatic activity of Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor
All present enzymatic activity of Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor:
3.1.3.11;
3.1.3.11;
Protein crystallography data
The structure of Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor, PDB code: 2fhy
was solved by
C.Abad-Zapatero,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 19.89 / 2.95 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 83.748, 109.126, 190.838, 90.00, 90.00, 90.00 |
| R / Rfree (%) | n/a / n/a |
Other elements in 2fhy:
The structure of Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor also contains other interesting chemical elements:
| Chlorine | (Cl) | 12 atoms |
Magnesium Binding Sites:
The binding sites of Magnesium atom in the Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor
(pdb code 2fhy). This binding sites where shown within
5.0 Angstroms radius around Magnesium atom.
In total 4 binding sites of Magnesium where determined in the Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor, PDB code: 2fhy:
Jump to Magnesium binding site number: 1; 2; 3; 4;
In total 4 binding sites of Magnesium where determined in the Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor, PDB code: 2fhy:
Jump to Magnesium binding site number: 1; 2; 3; 4;
Magnesium binding site 1 out of 4 in 2fhy
Go back to
Magnesium binding site 1 out
of 4 in the Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor
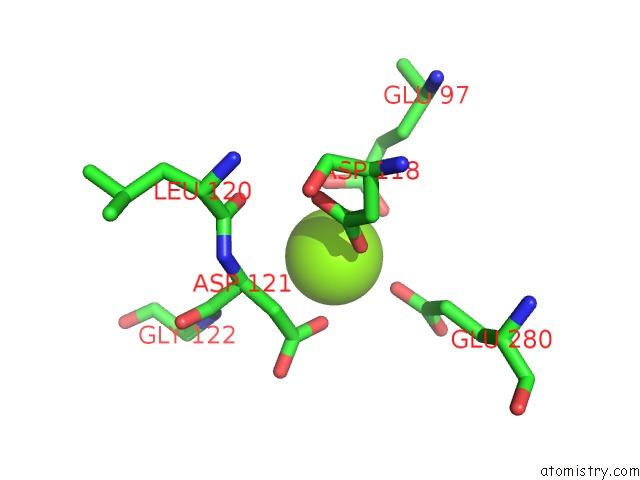
Mono view
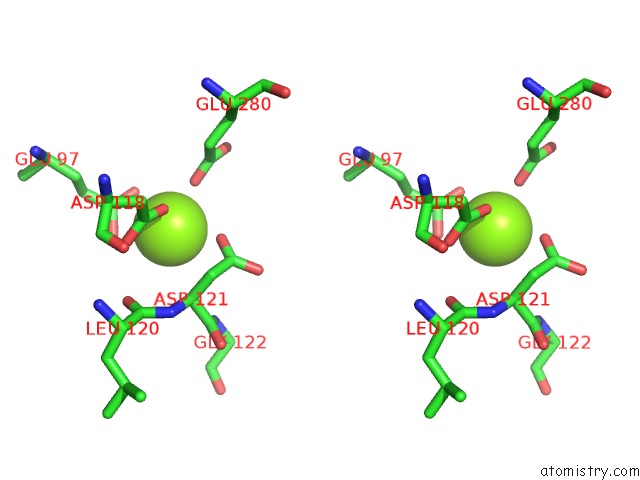
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 1 of Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor within 5.0Å range:
|
Magnesium binding site 2 out of 4 in 2fhy
Go back to
Magnesium binding site 2 out
of 4 in the Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor
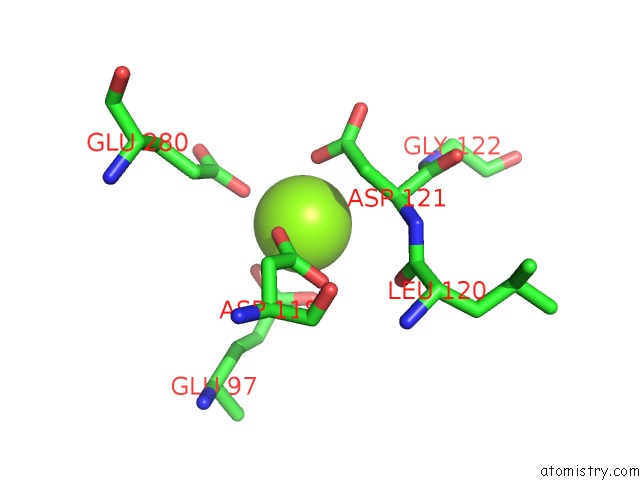
Mono view
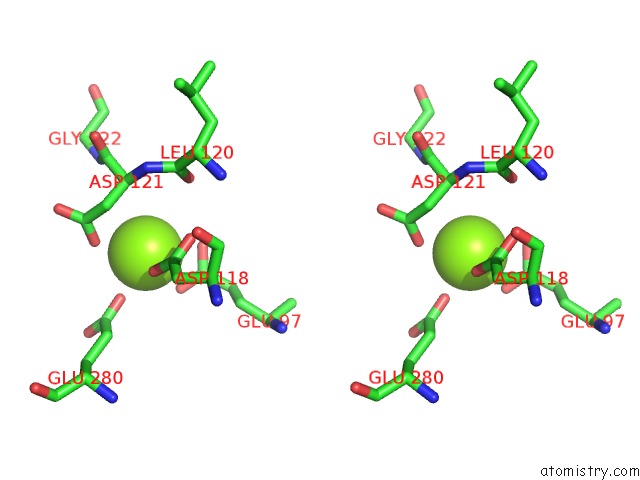
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 2 of Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor within 5.0Å range:
|
Magnesium binding site 3 out of 4 in 2fhy
Go back to
Magnesium binding site 3 out
of 4 in the Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor
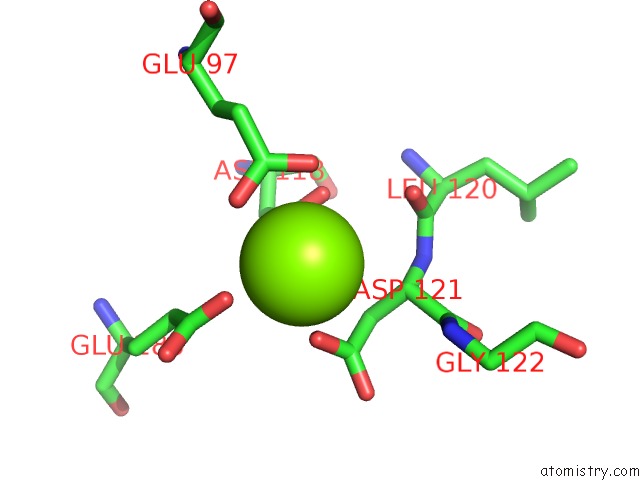
Mono view
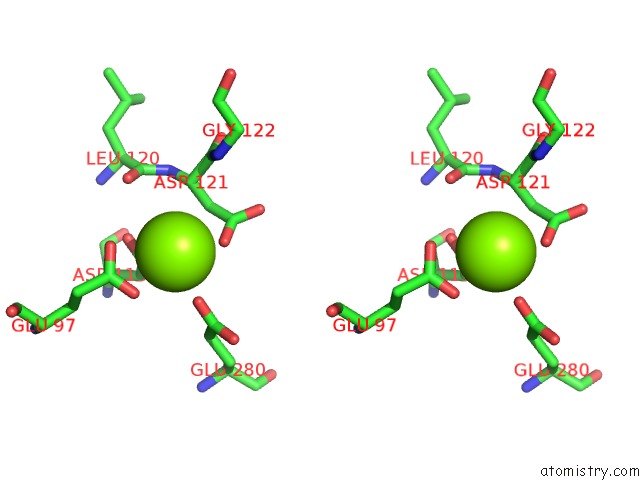
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 3 of Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor within 5.0Å range:
|
Magnesium binding site 4 out of 4 in 2fhy
Go back to
Magnesium binding site 4 out
of 4 in the Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor
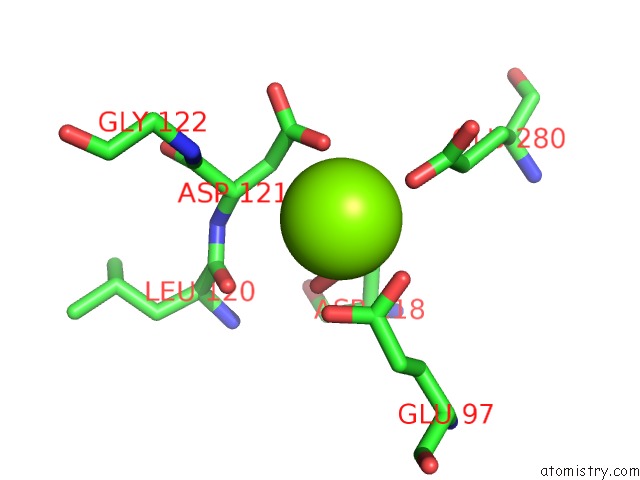
Mono view
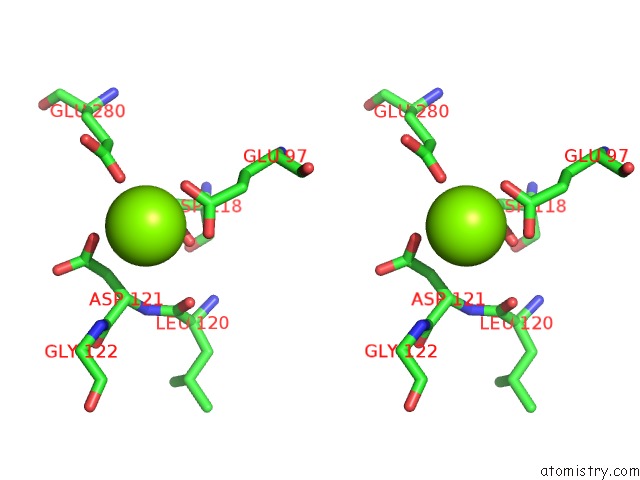
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 4 of Structure of Human Liver Fpbase Complexed with A Novel Benzoxazole As Allosteric Inhibitor within 5.0Å range:
|
Reference:
T.W.Von Geldern,
C.Lai,
R.J.Gum,
M.Daly,
C.Sun,
E.H.Fry,
C.Abad-Zapatero.
Benzoxazole Benzenesulfonamides Are Novel Allosteric Inhibitors of Fructose-1,6-Bisphosphatase with A Distinct Binding Mode. Bioorg.Med.Chem.Lett. V. 16 1811 2006.
ISSN: ISSN 0960-894X
PubMed: 16442285
DOI: 10.1016/J.BMCL.2006.01.015
Page generated: Tue Aug 13 23:07:13 2024
ISSN: ISSN 0960-894X
PubMed: 16442285
DOI: 10.1016/J.BMCL.2006.01.015
Last articles
F in 4KK8F in 4KJQ
F in 4KHM
F in 4KE5
F in 4KHR
F in 4KIL
F in 4KGJ
F in 4KFO
F in 4KH2
F in 4KBK