Magnesium »
PDB 3hx0-3i5x »
3hxy »
Magnesium in PDB 3hxy: Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp
Enzymatic activity of Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp
All present enzymatic activity of Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp:
6.1.1.7;
6.1.1.7;
Protein crystallography data
The structure of Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp, PDB code: 3hxy
was solved by
M.Guo,
X.-L.Yang,
P.Schimmel,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 41.56 / 2.27 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 44.355, 108.745, 118.985, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 18.1 / 22.7 |
Magnesium Binding Sites:
The binding sites of Magnesium atom in the Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp
(pdb code 3hxy). This binding sites where shown within
5.0 Angstroms radius around Magnesium atom.
In total 4 binding sites of Magnesium where determined in the Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp, PDB code: 3hxy:
Jump to Magnesium binding site number: 1; 2; 3; 4;
In total 4 binding sites of Magnesium where determined in the Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp, PDB code: 3hxy:
Jump to Magnesium binding site number: 1; 2; 3; 4;
Magnesium binding site 1 out of 4 in 3hxy
Go back to
Magnesium binding site 1 out
of 4 in the Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp
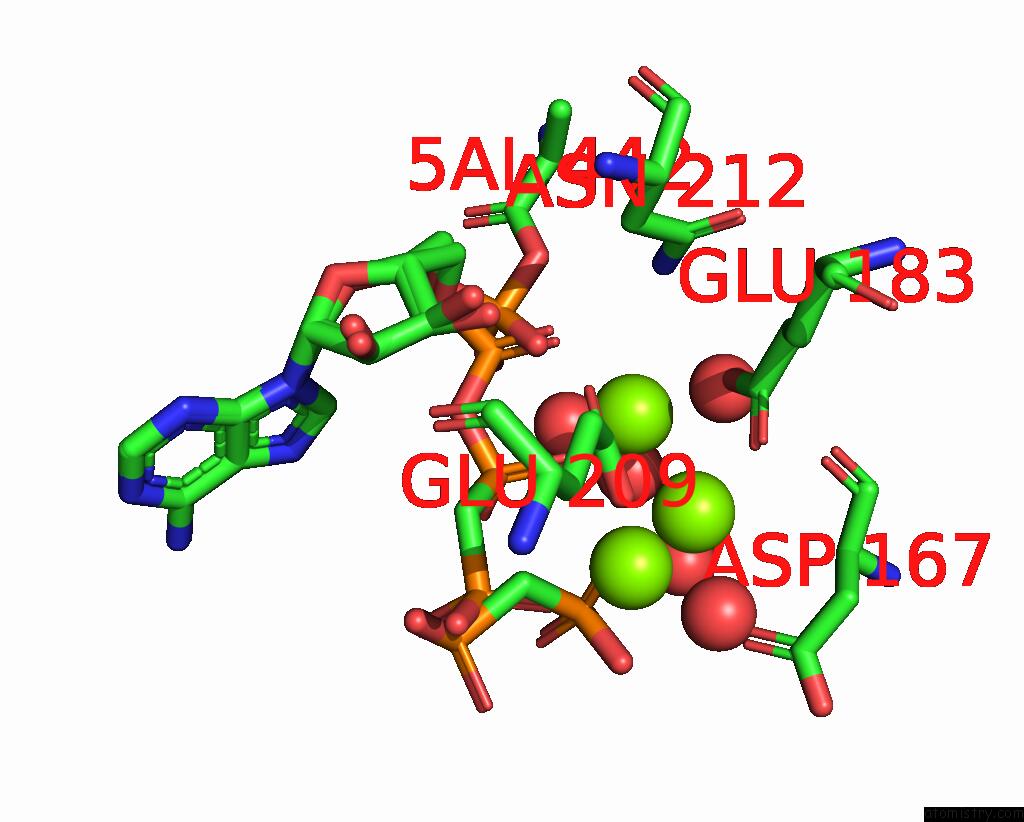
Mono view
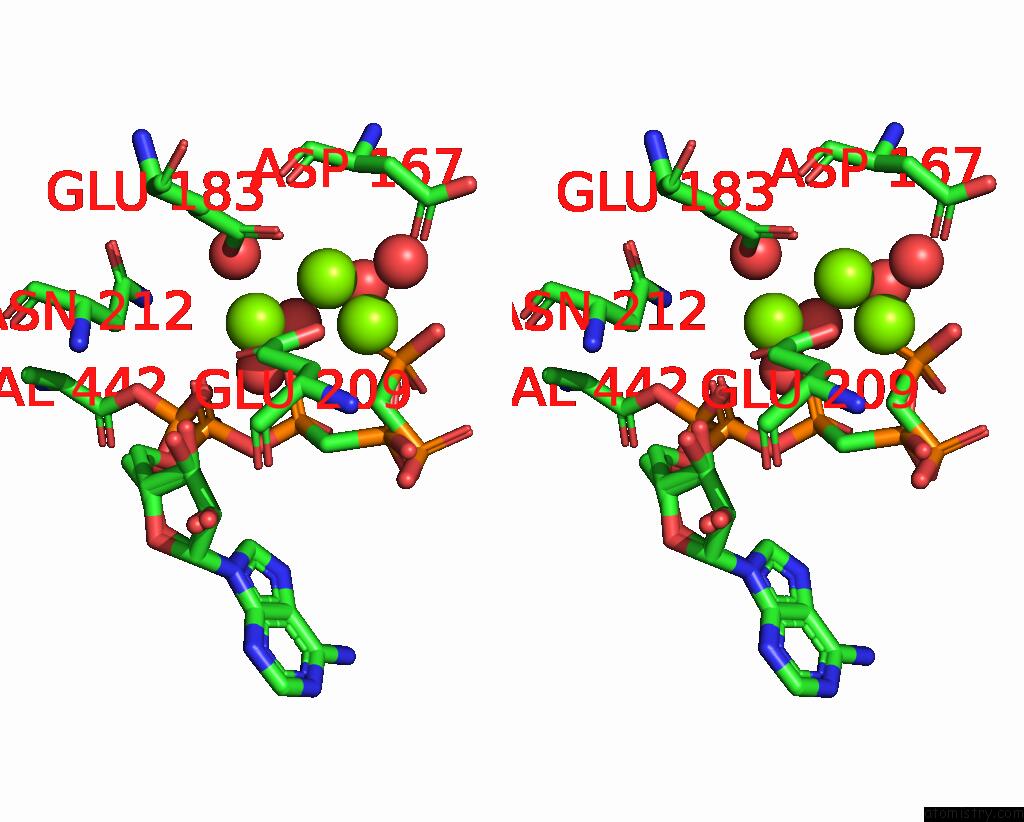
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 1 of Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp within 5.0Å range:
|
Magnesium binding site 2 out of 4 in 3hxy
Go back to
Magnesium binding site 2 out
of 4 in the Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp
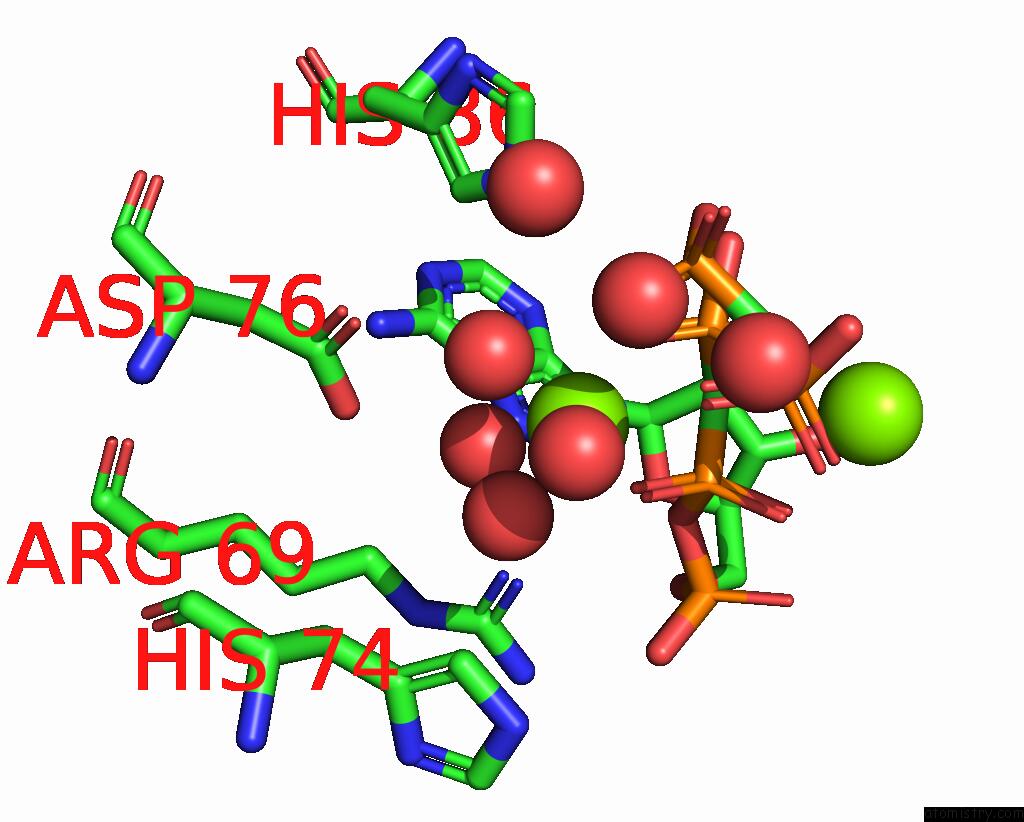
Mono view
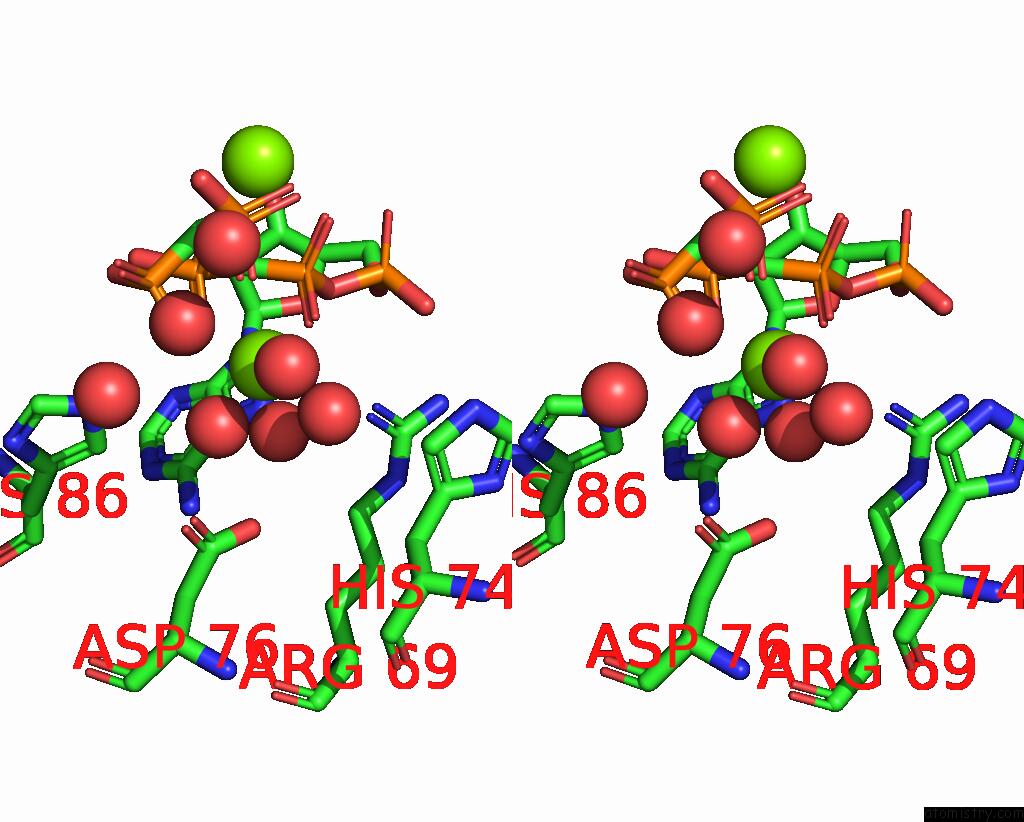
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 2 of Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp within 5.0Å range:
|
Magnesium binding site 3 out of 4 in 3hxy
Go back to
Magnesium binding site 3 out
of 4 in the Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp
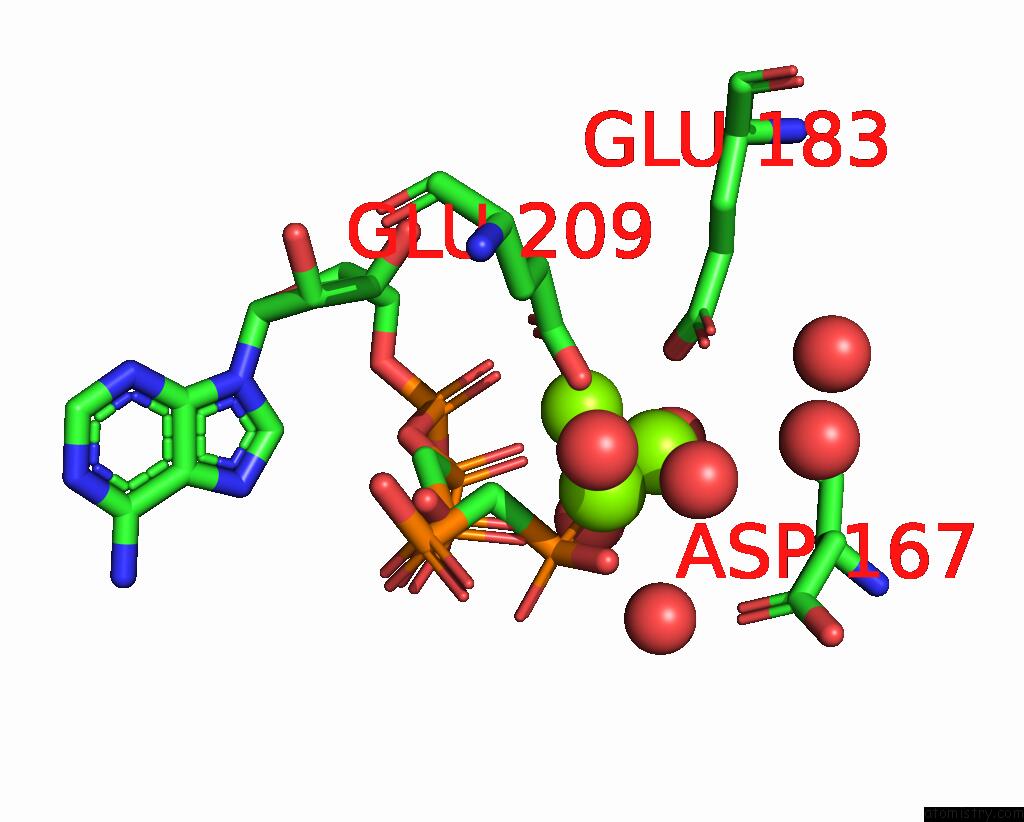
Mono view
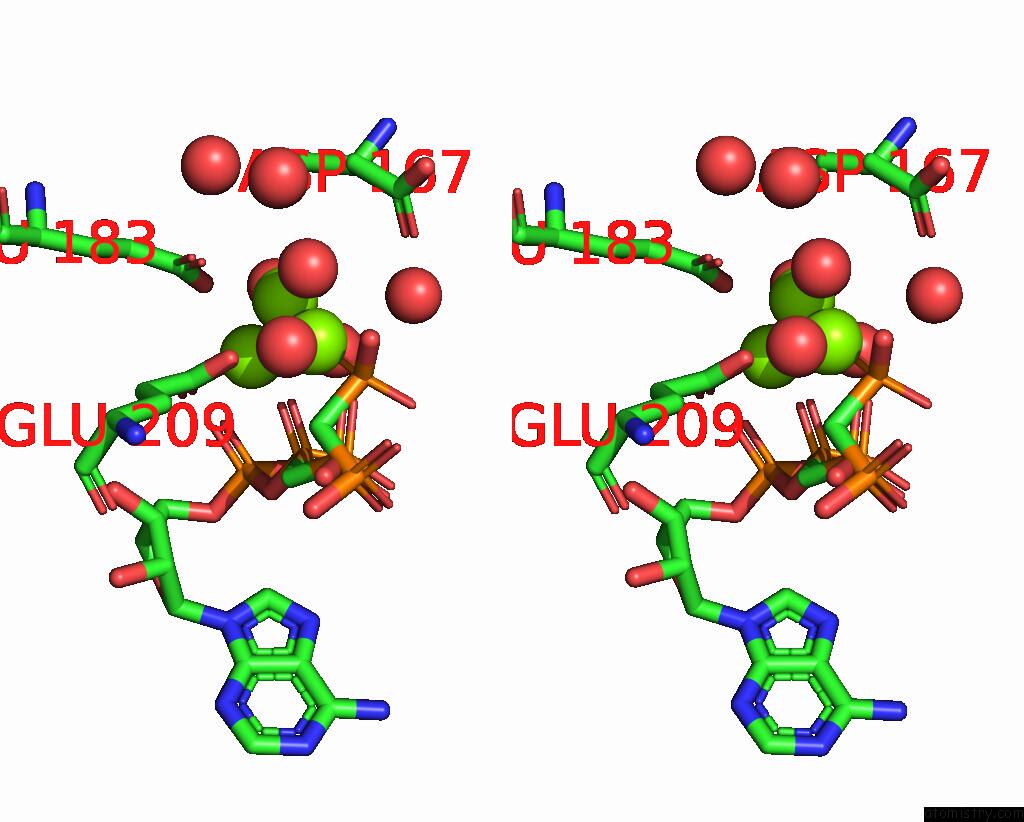
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 3 of Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp within 5.0Å range:
|
Magnesium binding site 4 out of 4 in 3hxy
Go back to
Magnesium binding site 4 out
of 4 in the Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp
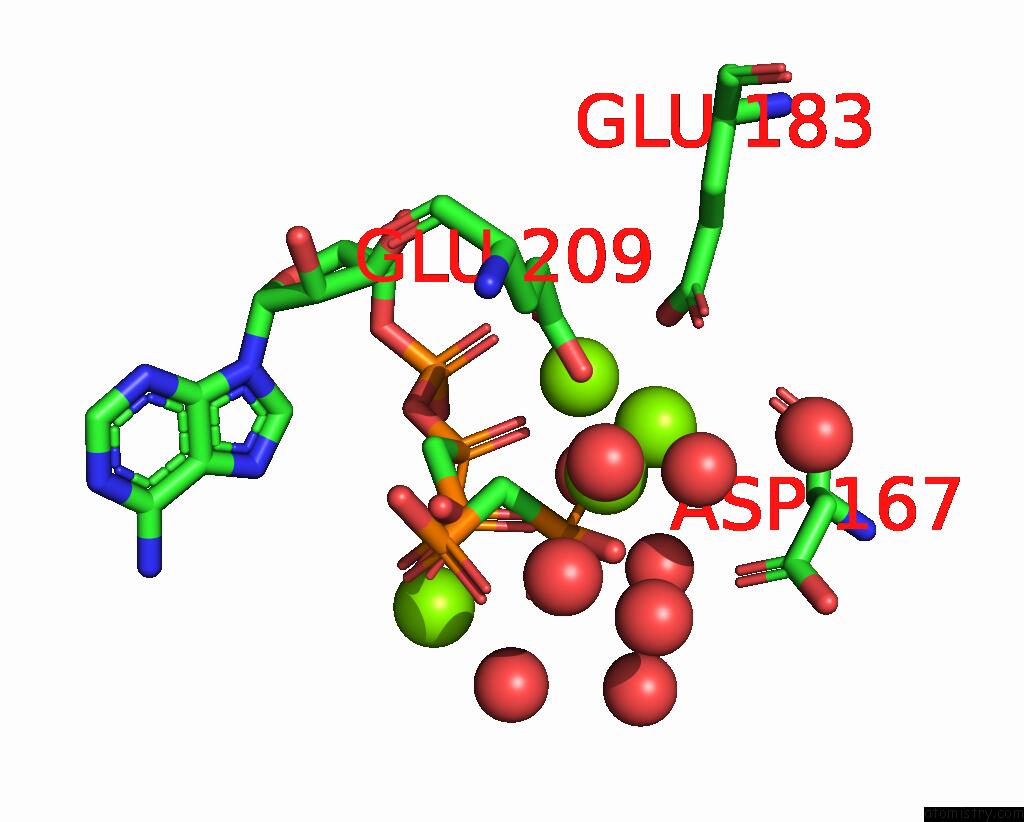
Mono view
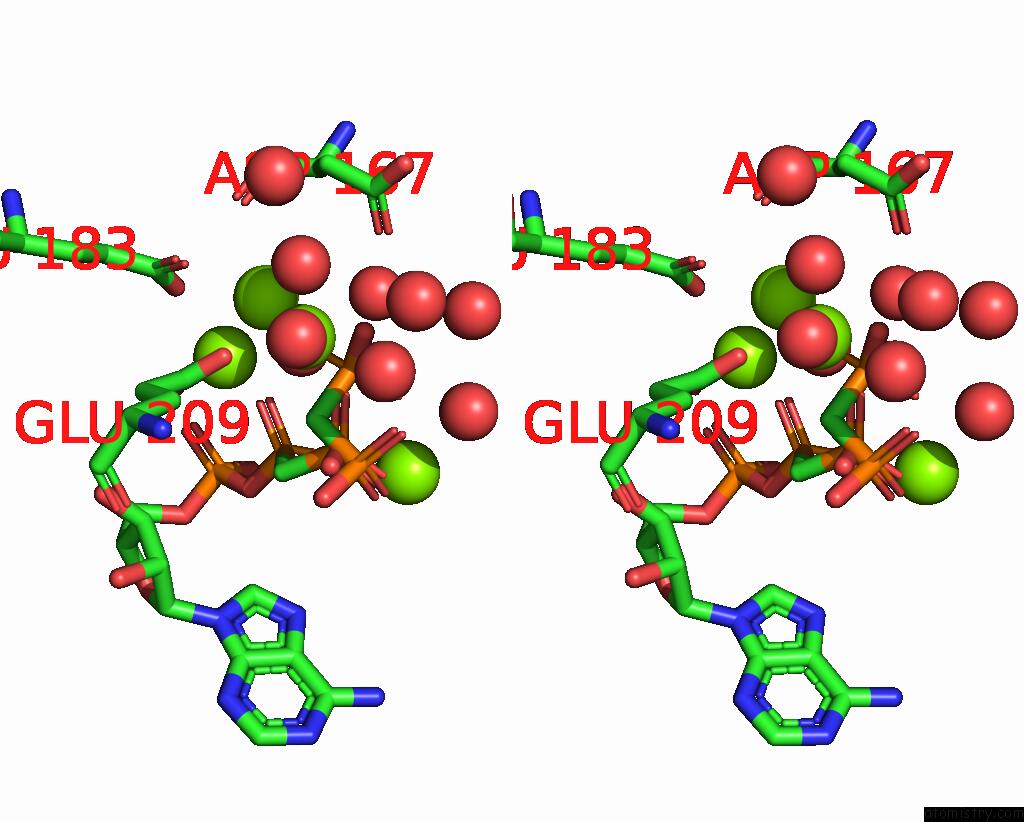
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 4 of Crystal Structure of Catalytic Fragment of E. Coli Alars in Complex with Amppcp, Ala-Amp and Pcp within 5.0Å range:
|
Reference:
M.Guo,
Y.E.Chong,
R.Shapiro,
K.Beebe,
X.L.Yang,
P.Schimmel.
Paradox of Mistranslation of Serine For Alanine Caused By Alars Recognition Dilemma. Nature V. 462 808 2009.
ISSN: ISSN 0028-0836
PubMed: 20010690
DOI: 10.1038/NATURE08612
Page generated: Sun Aug 10 22:09:16 2025
ISSN: ISSN 0028-0836
PubMed: 20010690
DOI: 10.1038/NATURE08612
Last articles
Mg in 4APZMg in 4AV6
Mg in 4AVA
Mg in 4AV3
Mg in 4AUX
Mg in 4ATB
Mg in 4AUI
Mg in 4AT9
Mg in 4AT8
Mg in 4AS5