Magnesium »
PDB 3oto-3p0x »
3p0x »
Magnesium in PDB 3p0x: Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate
Enzymatic activity of Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate
All present enzymatic activity of Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate:
4.1.3.1;
4.1.3.1;
Protein crystallography data
The structure of Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate, PDB code: 3p0x
was solved by
Seattle Structural Genomics Center For Infectious Disease (Ssgcid),
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 47.60 / 2.35 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 76.910, 135.930, 181.790, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 17.8 / 23 |
Magnesium Binding Sites:
The binding sites of Magnesium atom in the Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate
(pdb code 3p0x). This binding sites where shown within
5.0 Angstroms radius around Magnesium atom.
In total 4 binding sites of Magnesium where determined in the Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate, PDB code: 3p0x:
Jump to Magnesium binding site number: 1; 2; 3; 4;
In total 4 binding sites of Magnesium where determined in the Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate, PDB code: 3p0x:
Jump to Magnesium binding site number: 1; 2; 3; 4;
Magnesium binding site 1 out of 4 in 3p0x
Go back to
Magnesium binding site 1 out
of 4 in the Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate
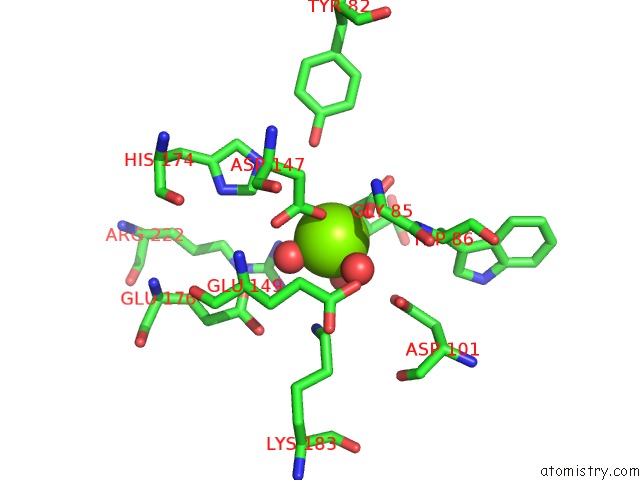
Mono view
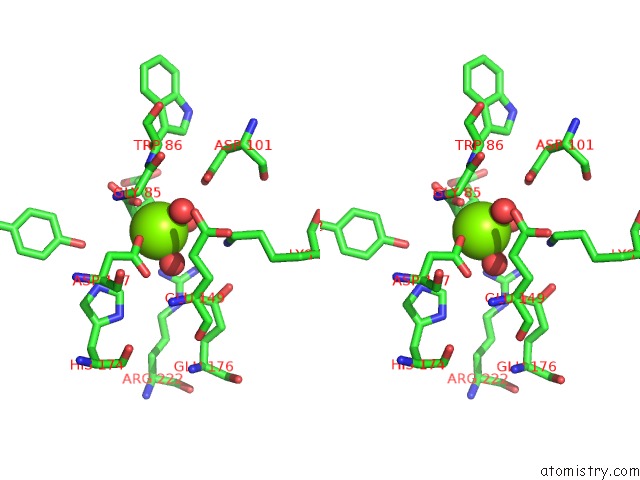
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 1 of Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate within 5.0Å range:
|
Magnesium binding site 2 out of 4 in 3p0x
Go back to
Magnesium binding site 2 out
of 4 in the Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate
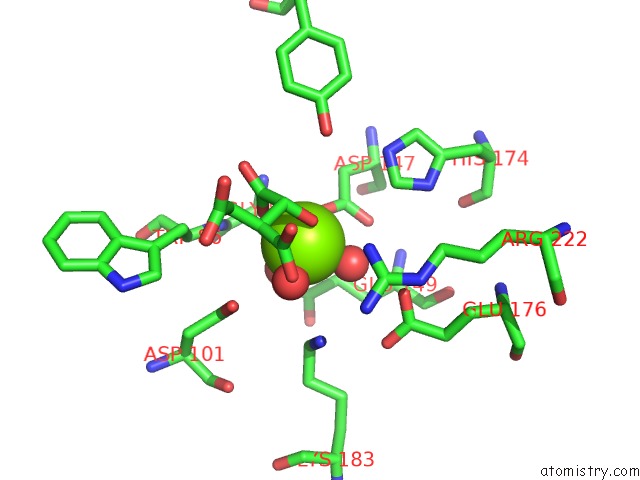
Mono view
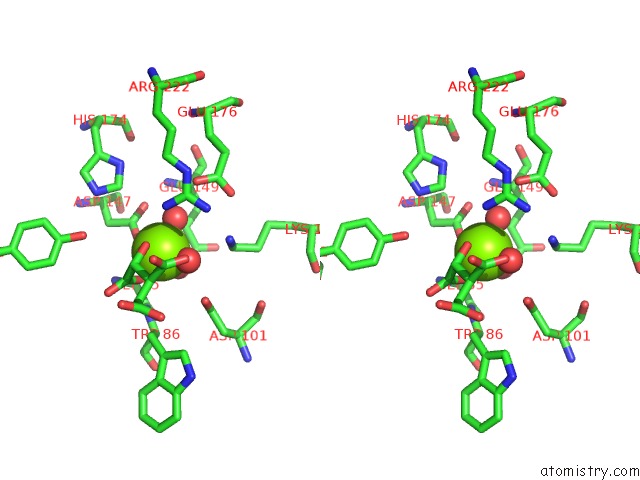
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 2 of Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate within 5.0Å range:
|
Magnesium binding site 3 out of 4 in 3p0x
Go back to
Magnesium binding site 3 out
of 4 in the Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate
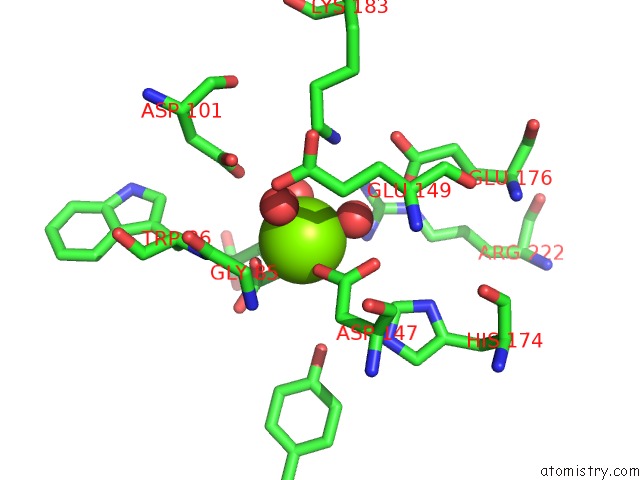
Mono view
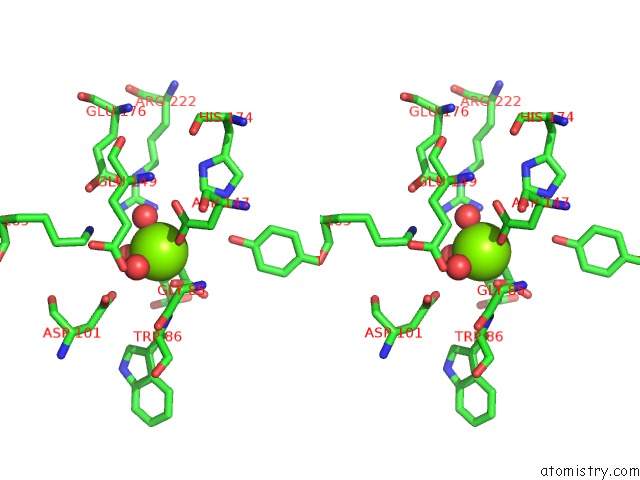
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 3 of Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate within 5.0Å range:
|
Magnesium binding site 4 out of 4 in 3p0x
Go back to
Magnesium binding site 4 out
of 4 in the Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate
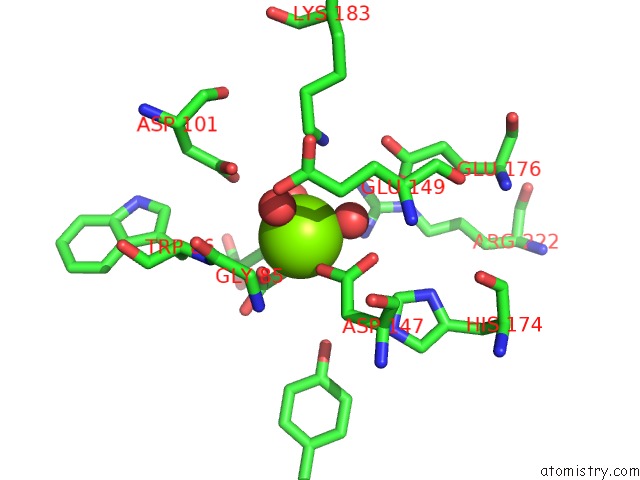
Mono view
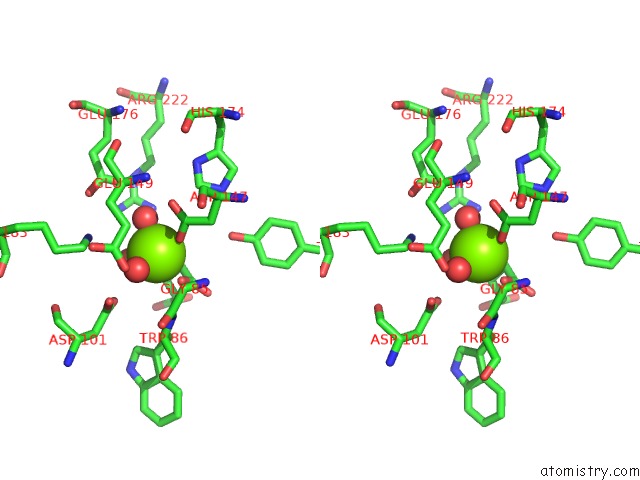
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 4 of Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate within 5.0Å range:
|
Reference:
Ssgcid,
A.Gardberg,
T.Edwards,
M.Gander,
B.Staker,
L.Stewart.
Crystal Structure of Isocitrate Lyase From Brucella Melitensis, Bound to Magnesium Isocitrate To Be Published.
Page generated: Thu Aug 15 09:02:17 2024
Last articles
Cl in 7UUJCl in 7UUG
Cl in 7USZ
Cl in 7UP4
Cl in 7UOS
Cl in 7UPI
Cl in 7UMO
Cl in 7UOD
Cl in 7UN0
Cl in 7UOQ