Magnesium »
PDB 3py8-3q86 »
3q7u »
Magnesium in PDB 3q7u: Structure of Mtb 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd) Complexed with Ctp
Enzymatic activity of Structure of Mtb 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd) Complexed with Ctp
All present enzymatic activity of Structure of Mtb 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd) Complexed with Ctp:
2.7.7.60;
2.7.7.60;
Protein crystallography data
The structure of Structure of Mtb 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd) Complexed with Ctp, PDB code: 3q7u
was solved by
M.C.M.Reddy,
J.B.Bruning,
C.Thurman,
T.R.Ioerger,
J.C.Sacchettini,
Tbstructural Genomics Consortium (Tbsgc),
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 38.38 / 2.10 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 55.214, 76.756, 131.538, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 18.8 / 23.6 |
Magnesium Binding Sites:
The binding sites of Magnesium atom in the Structure of Mtb 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd) Complexed with Ctp
(pdb code 3q7u). This binding sites where shown within
5.0 Angstroms radius around Magnesium atom.
In total 2 binding sites of Magnesium where determined in the Structure of Mtb 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd) Complexed with Ctp, PDB code: 3q7u:
Jump to Magnesium binding site number: 1; 2;
In total 2 binding sites of Magnesium where determined in the Structure of Mtb 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd) Complexed with Ctp, PDB code: 3q7u:
Jump to Magnesium binding site number: 1; 2;
Magnesium binding site 1 out of 2 in 3q7u
Go back to
Magnesium binding site 1 out
of 2 in the Structure of Mtb 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd) Complexed with Ctp
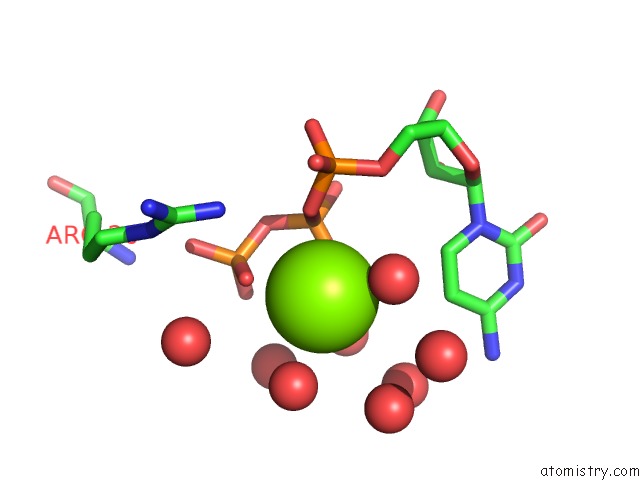
Mono view
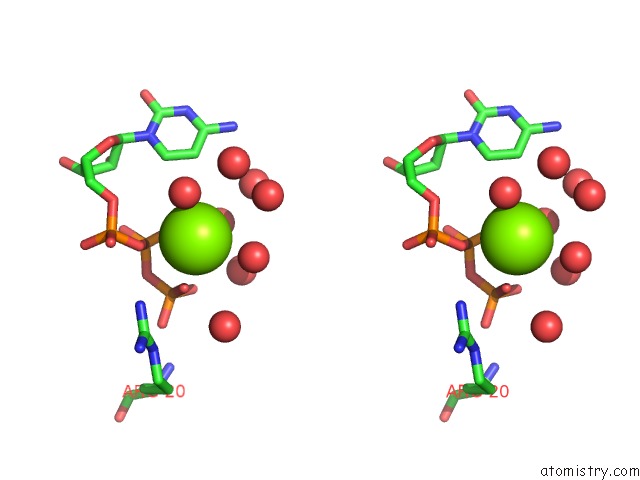
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 1 of Structure of Mtb 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd) Complexed with Ctp within 5.0Å range:
|
Magnesium binding site 2 out of 2 in 3q7u
Go back to
Magnesium binding site 2 out
of 2 in the Structure of Mtb 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd) Complexed with Ctp
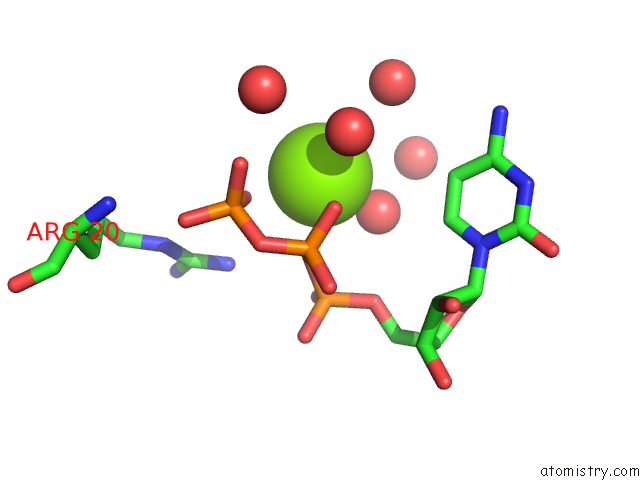
Mono view
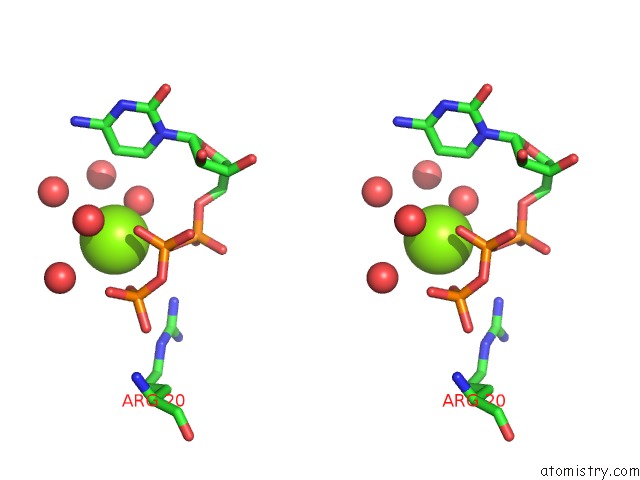
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 2 of Structure of Mtb 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd) Complexed with Ctp within 5.0Å range:
|
Reference:
M.C.M.Reddy,
J.B.Bruning,
C.Thurman,
T.R.Ioerger,
J.C.Sacchettini.
Crystal Structure of Mycobacterium Tuberculosis 2-C-Methyl-D-Erythritol 4-Phosphate Cytidyltransferase (Ispd): A Candidate Antitubercular Drug Target Proteins 2011.
ISSN: ESSN 1097-0134
Page generated: Thu Aug 15 09:58:45 2024
ISSN: ESSN 1097-0134
Last articles
Ca in 5SZLCa in 5SY1
Ca in 5SWI
Ca in 5SVE
Ca in 5SSX
Ca in 5SV0
Ca in 5STD
Ca in 5SSZ
Ca in 5SSY
Ca in 5SIC