Magnesium »
PDB 5grf-5h2f »
5gul »
Magnesium in PDB 5gul: Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190
Protein crystallography data
The structure of Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190, PDB code: 5gul
was solved by
T.Tomita,
M.Kobayashi,
M.Nishiyama,
T.Kuzuyama,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 55.04 / 1.73 |
| Space group | P 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 46.851, 61.661, 82.242, 74.00, 84.54, 86.04 |
| R / Rfree (%) | 21.6 / 27.8 |
Magnesium Binding Sites:
The binding sites of Magnesium atom in the Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190
(pdb code 5gul). This binding sites where shown within
5.0 Angstroms radius around Magnesium atom.
In total 4 binding sites of Magnesium where determined in the Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190, PDB code: 5gul:
Jump to Magnesium binding site number: 1; 2; 3; 4;
In total 4 binding sites of Magnesium where determined in the Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190, PDB code: 5gul:
Jump to Magnesium binding site number: 1; 2; 3; 4;
Magnesium binding site 1 out of 4 in 5gul
Go back to
Magnesium binding site 1 out
of 4 in the Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190
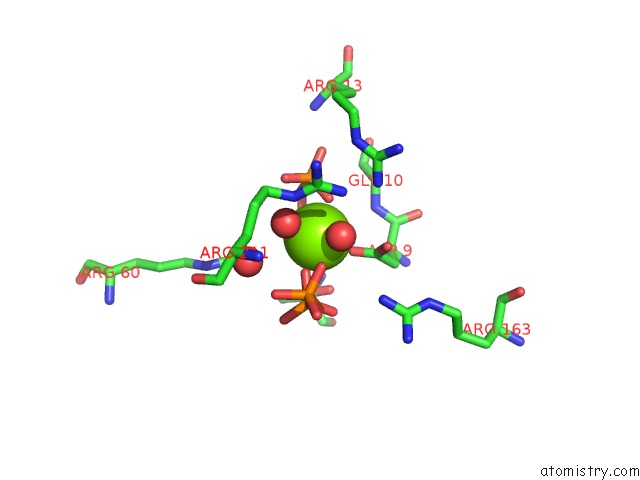
Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 1 of Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190 within 5.0Å range:
|
Magnesium binding site 2 out of 4 in 5gul
Go back to
Magnesium binding site 2 out
of 4 in the Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190

Mono view
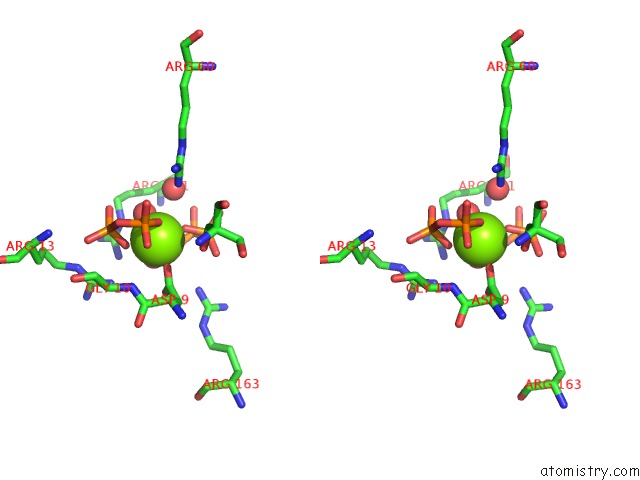
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 2 of Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190 within 5.0Å range:
|
Magnesium binding site 3 out of 4 in 5gul
Go back to
Magnesium binding site 3 out
of 4 in the Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190
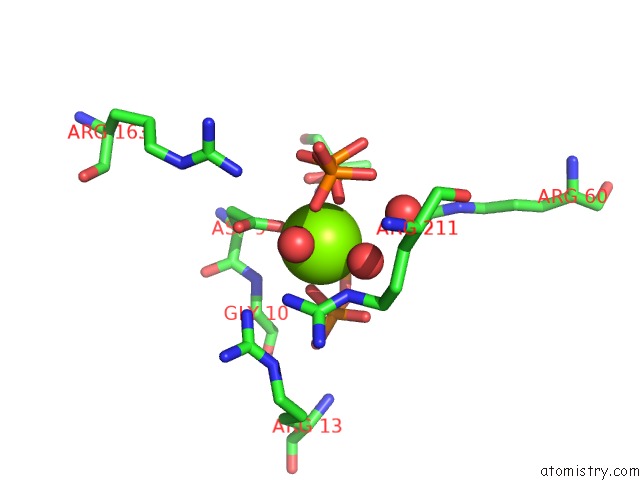
Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 3 of Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190 within 5.0Å range:
|
Magnesium binding site 4 out of 4 in 5gul
Go back to
Magnesium binding site 4 out
of 4 in the Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190
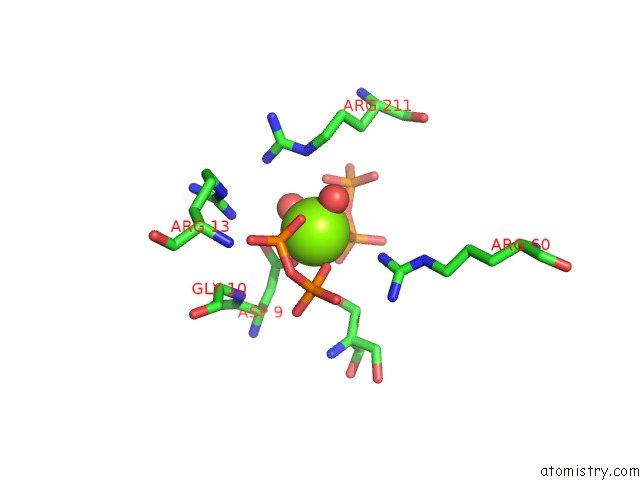
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 4 of Crystal Structure of Tris/PPIX2/MG2+ Bound Form of Cyclolavandulyl Diphosphate Synthase (Clds) From Streptomyces Sp. CL190 within 5.0Å range:
|
Reference:
T.Tomita,
M.Kobayashi,
Y.Karita,
Y.Yasuno,
T.Shinada,
M.Nishiyama,
T.Kuzuyama.
Structure and Mechanism of the Monoterpene Cyclolavandulyl Diphosphate Synthase That Catalyzes Consecutive Condensation and Cyclization. Angew. Chem. Int. Ed. Engl. V. 56 14913 2017.
ISSN: ESSN 1521-3773
PubMed: 28922556
DOI: 10.1002/ANIE.201708474
Page generated: Sun Sep 29 15:26:27 2024
ISSN: ESSN 1521-3773
PubMed: 28922556
DOI: 10.1002/ANIE.201708474
Last articles
Fe in 2YXOFe in 2YRS
Fe in 2YXC
Fe in 2YNM
Fe in 2YVJ
Fe in 2YP1
Fe in 2YU2
Fe in 2YU1
Fe in 2YQB
Fe in 2YOO