Magnesium »
PDB 5s60-5scf »
5scd »
Magnesium in PDB 5scd: Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58
Enzymatic activity of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58
All present enzymatic activity of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58:
2.7.1.40;
2.7.1.40;
Protein crystallography data
The structure of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58, PDB code: 5scd
was solved by
A.Lulla,
A.Foller,
A.Nain-Perez,
M.Grotli,
P.Brear,
M.Hyvonen,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 103.82 / 2.04 |
| Space group | C 1 2 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 207.725, 113.012, 188.817, 90, 91.63, 90 |
| R / Rfree (%) | 20.2 / 22.6 |
Other elements in 5scd:
The structure of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58 also contains other interesting chemical elements:
| Potassium | (K) | 8 atoms |
Magnesium Binding Sites:
The binding sites of Magnesium atom in the Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58
(pdb code 5scd). This binding sites where shown within
5.0 Angstroms radius around Magnesium atom.
In total 8 binding sites of Magnesium where determined in the Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58, PDB code: 5scd:
Jump to Magnesium binding site number: 1; 2; 3; 4; 5; 6; 7; 8;
In total 8 binding sites of Magnesium where determined in the Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58, PDB code: 5scd:
Jump to Magnesium binding site number: 1; 2; 3; 4; 5; 6; 7; 8;
Magnesium binding site 1 out of 8 in 5scd
Go back to
Magnesium binding site 1 out
of 8 in the Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58
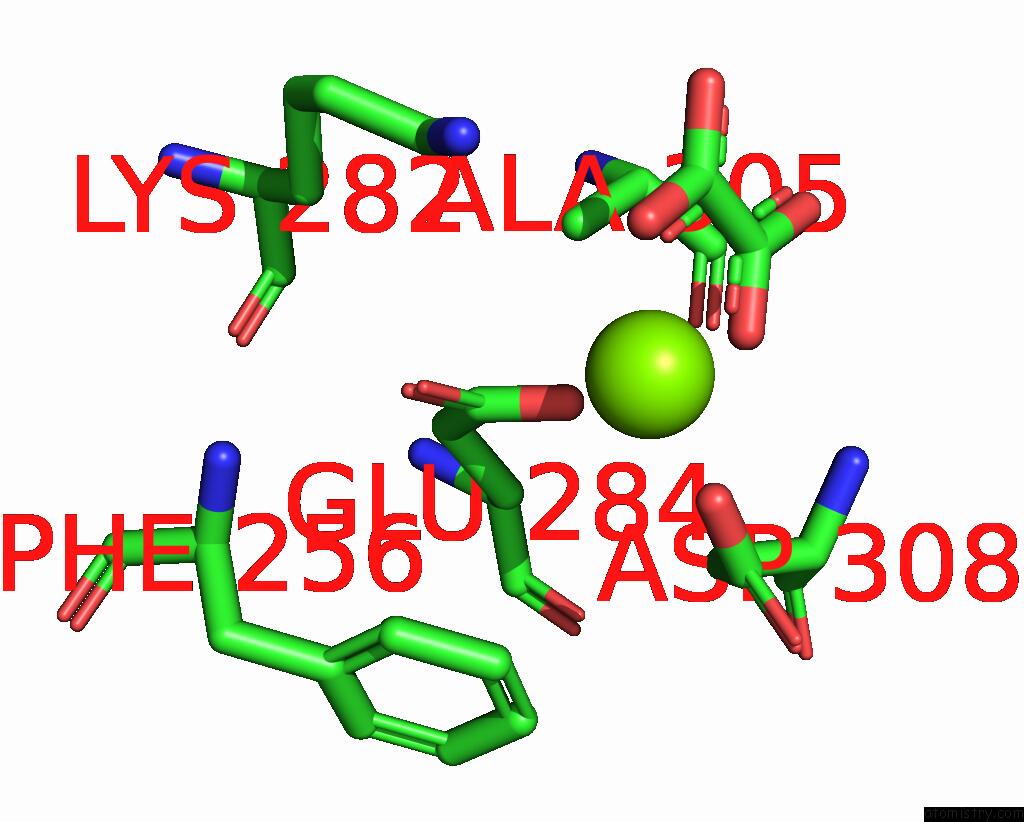
Mono view
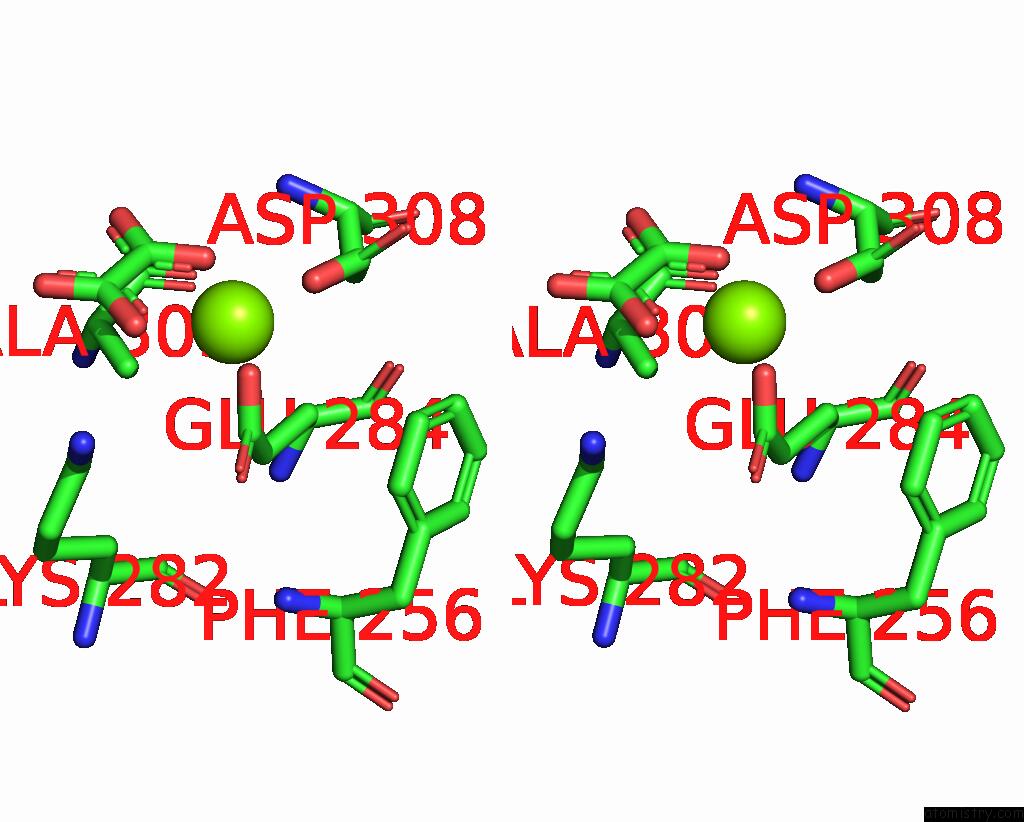
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 1 of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58 within 5.0Å range:
|
Magnesium binding site 2 out of 8 in 5scd
Go back to
Magnesium binding site 2 out
of 8 in the Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58
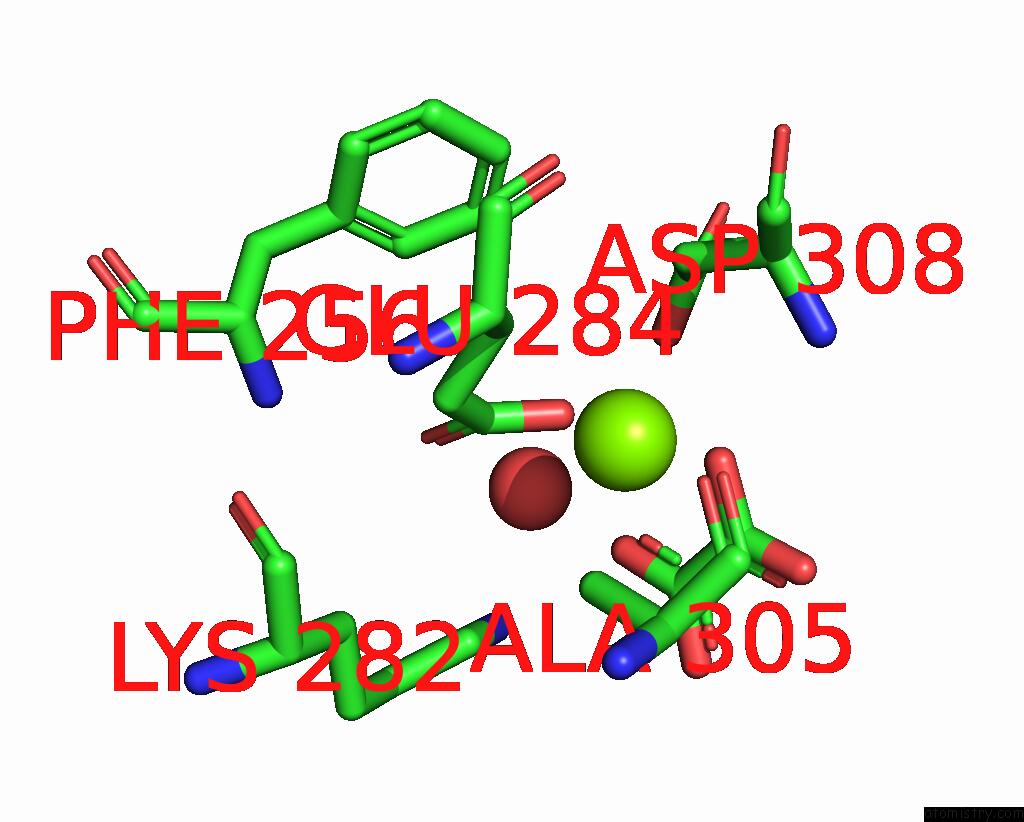
Mono view
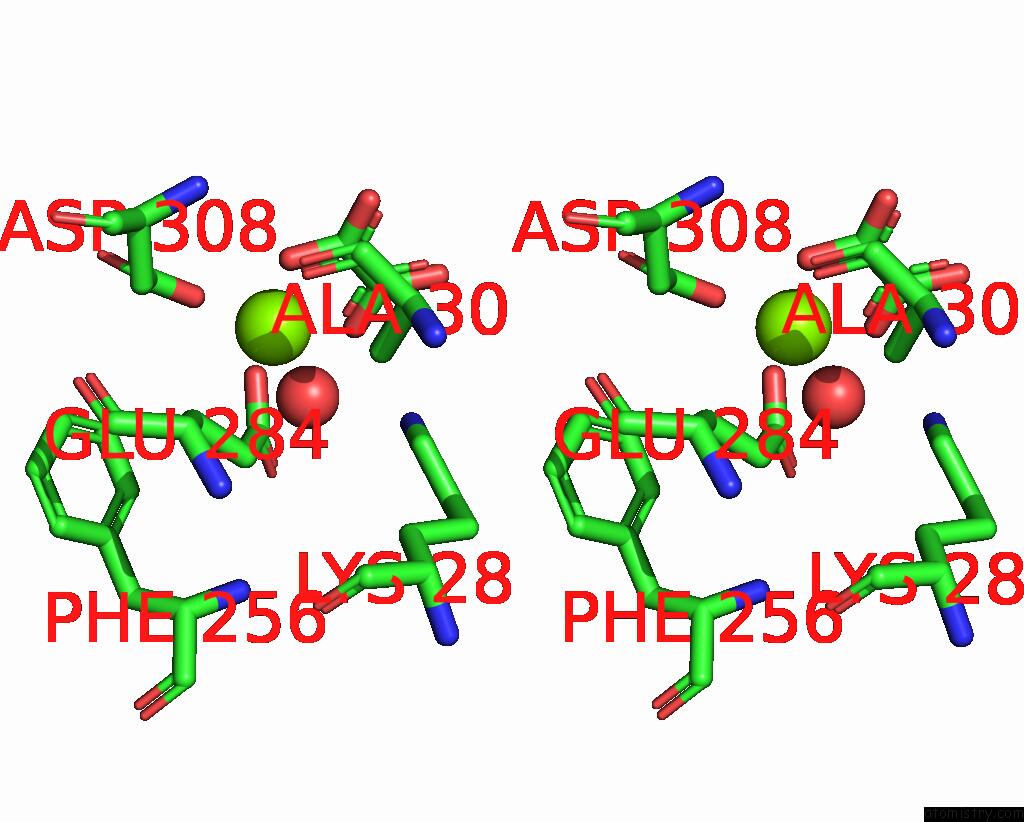
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 2 of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58 within 5.0Å range:
|
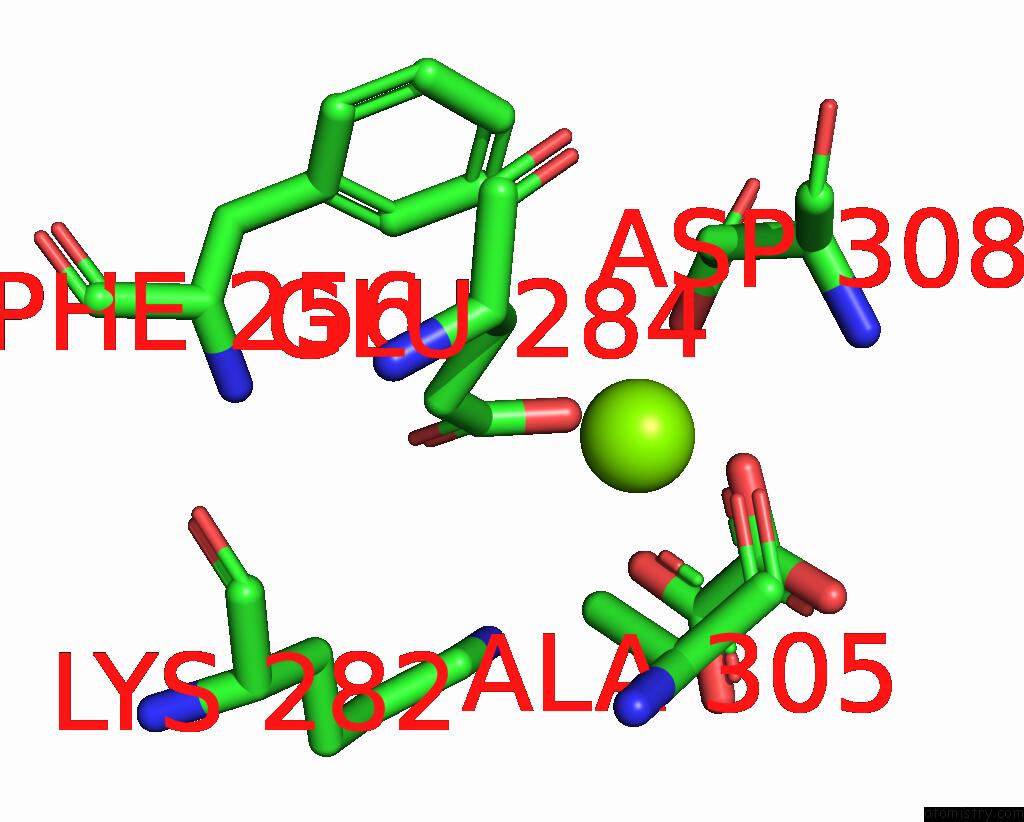
Magnesium binding site 3 out of 8 in 5scd
Go back to
Magnesium binding site 3 out
of 8 in the Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58
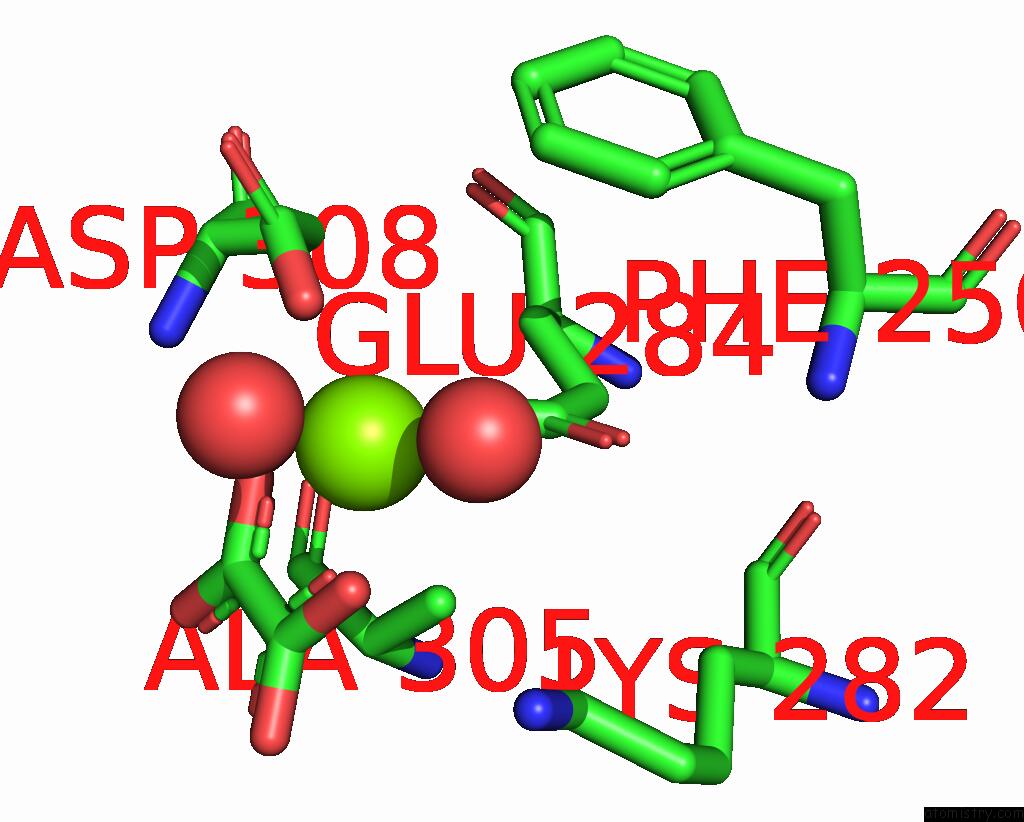
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 3 of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58 within 5.0Å range:
|
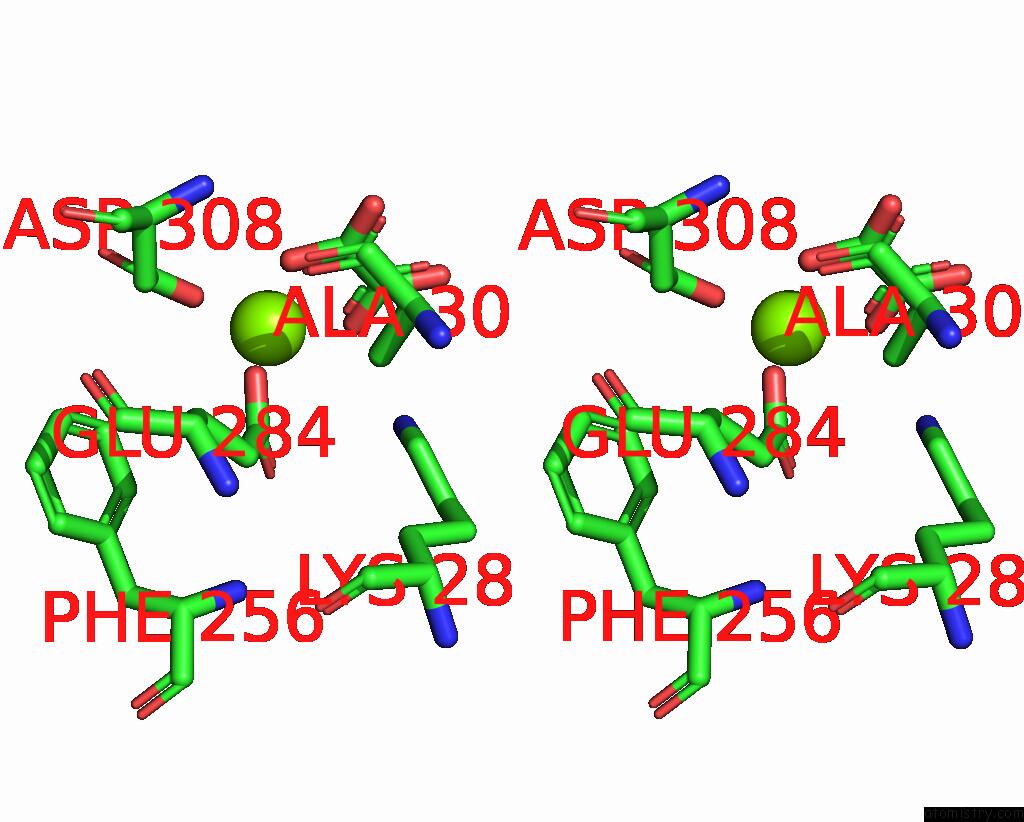
Magnesium binding site 4 out of 8 in 5scd
Go back to
Magnesium binding site 4 out
of 8 in the Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58
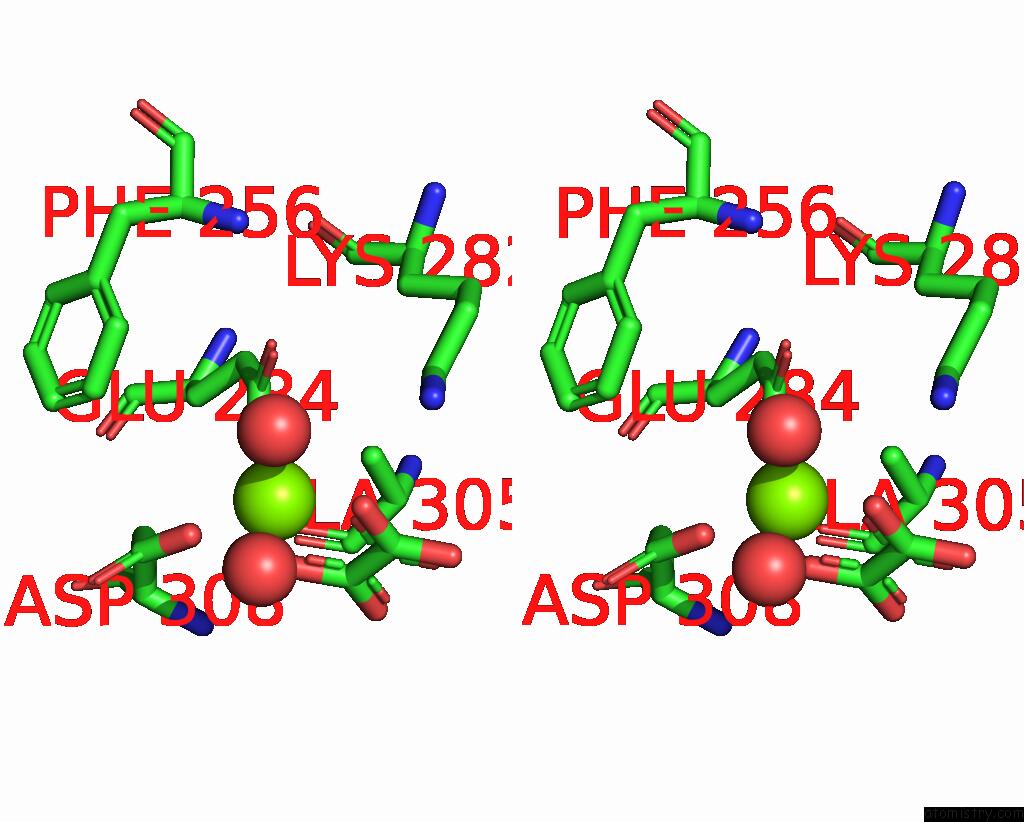
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 4 of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58 within 5.0Å range:
|
Magnesium binding site 5 out of 8 in 5scd
Go back to
Magnesium binding site 5 out
of 8 in the Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58
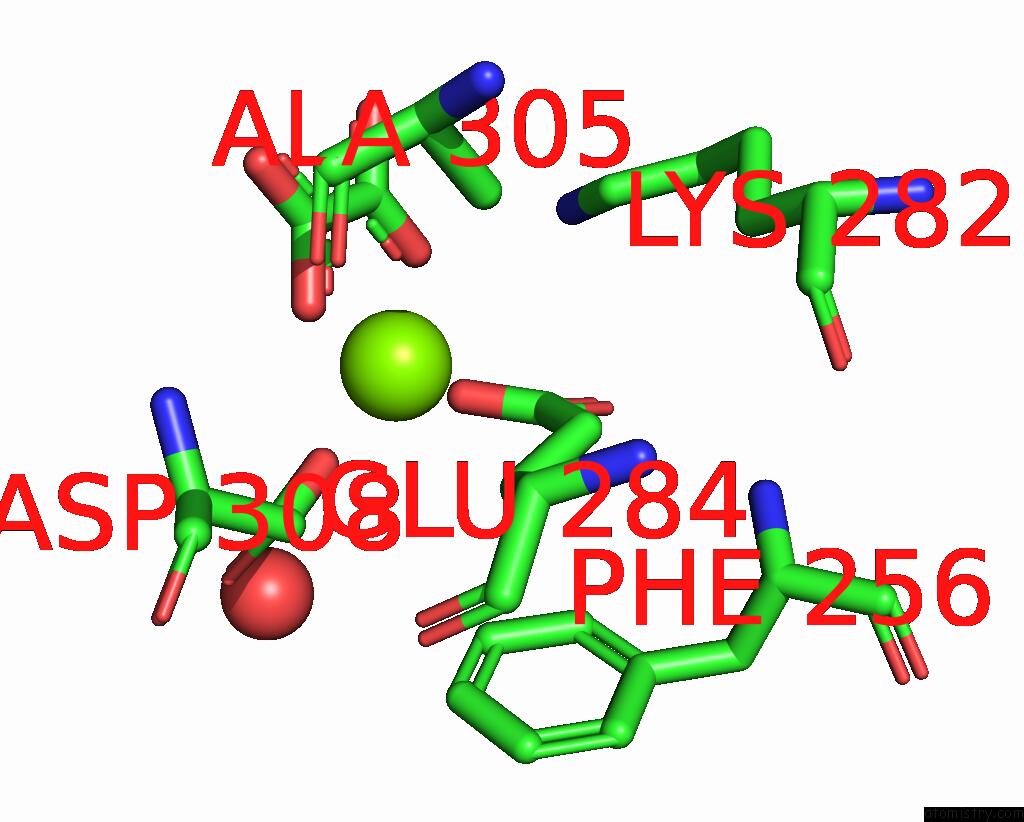
Mono view
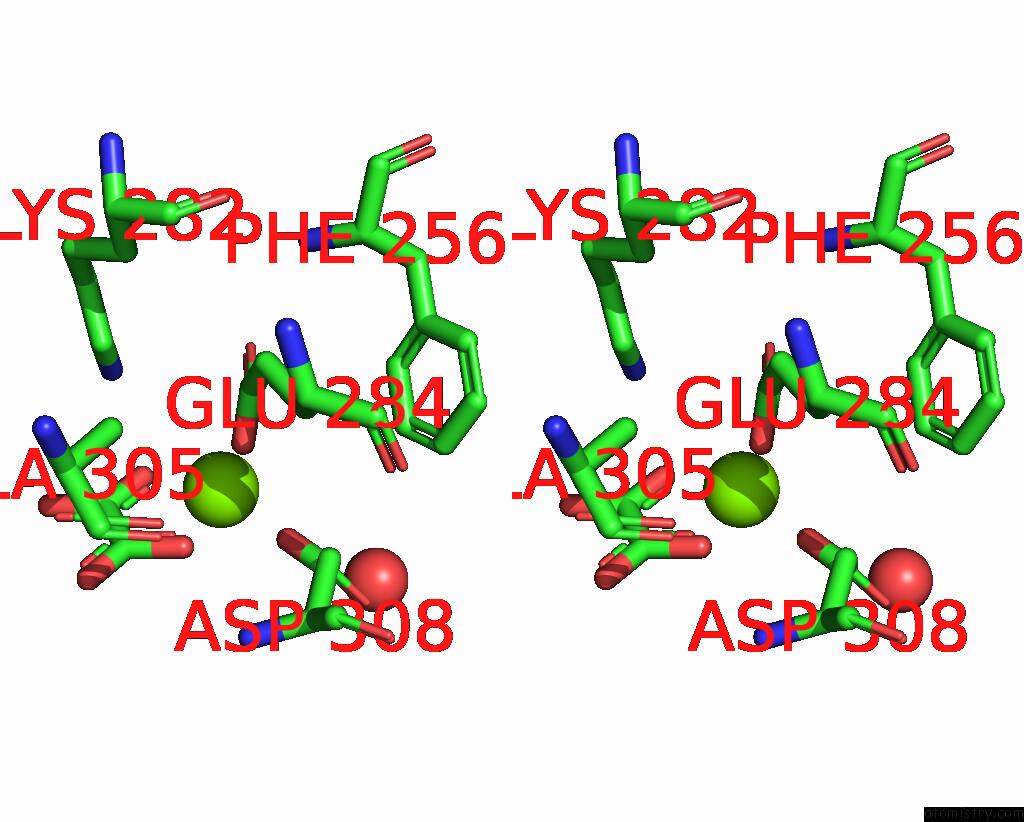
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 5 of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58 within 5.0Å range:
|
Magnesium binding site 6 out of 8 in 5scd
Go back to
Magnesium binding site 6 out
of 8 in the Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58
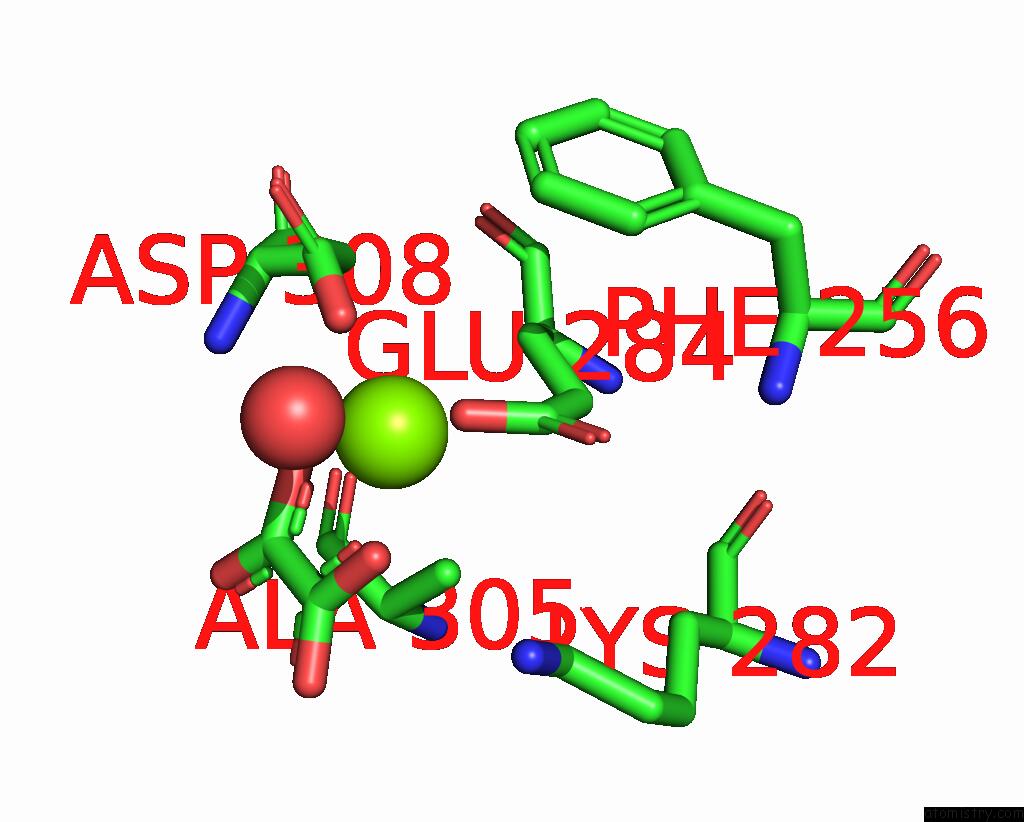
Mono view
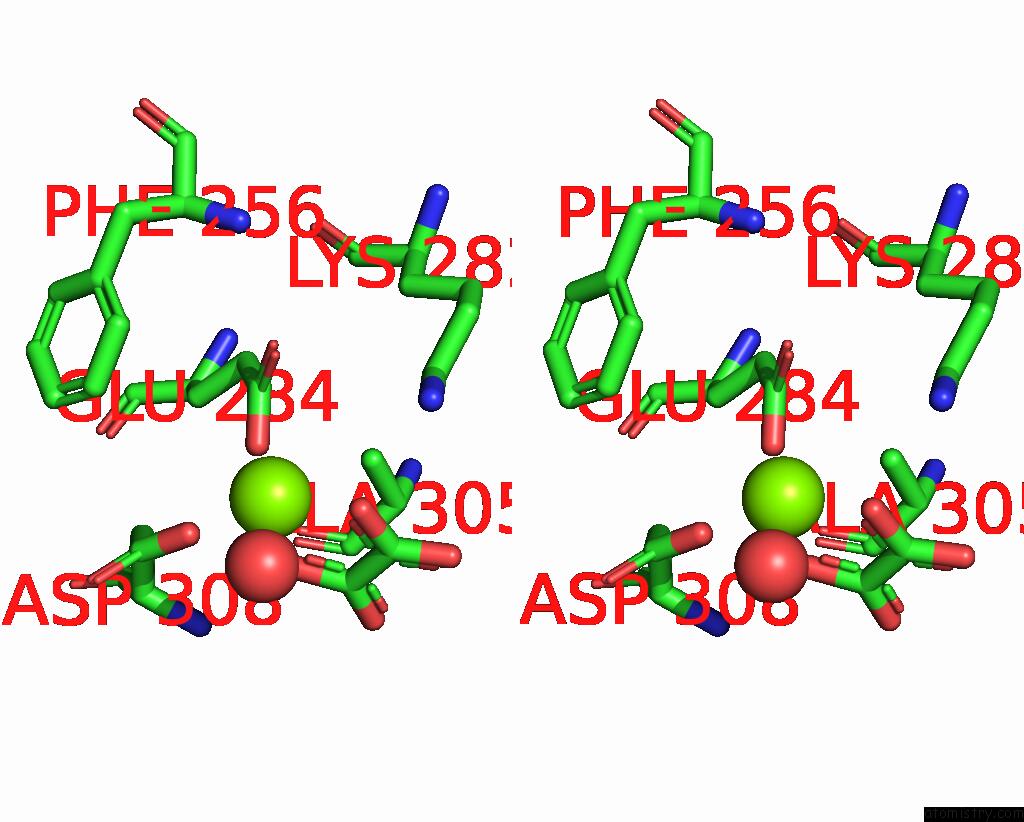
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 6 of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58 within 5.0Å range:
|
Magnesium binding site 7 out of 8 in 5scd
Go back to
Magnesium binding site 7 out
of 8 in the Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58
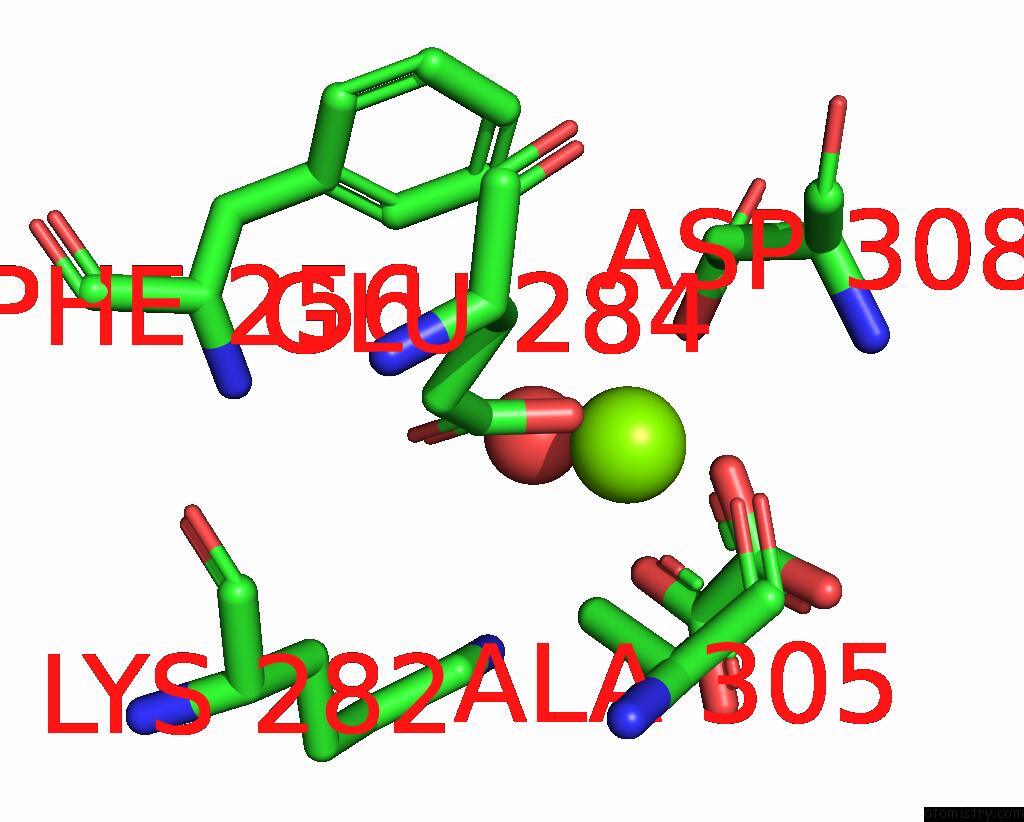
Mono view
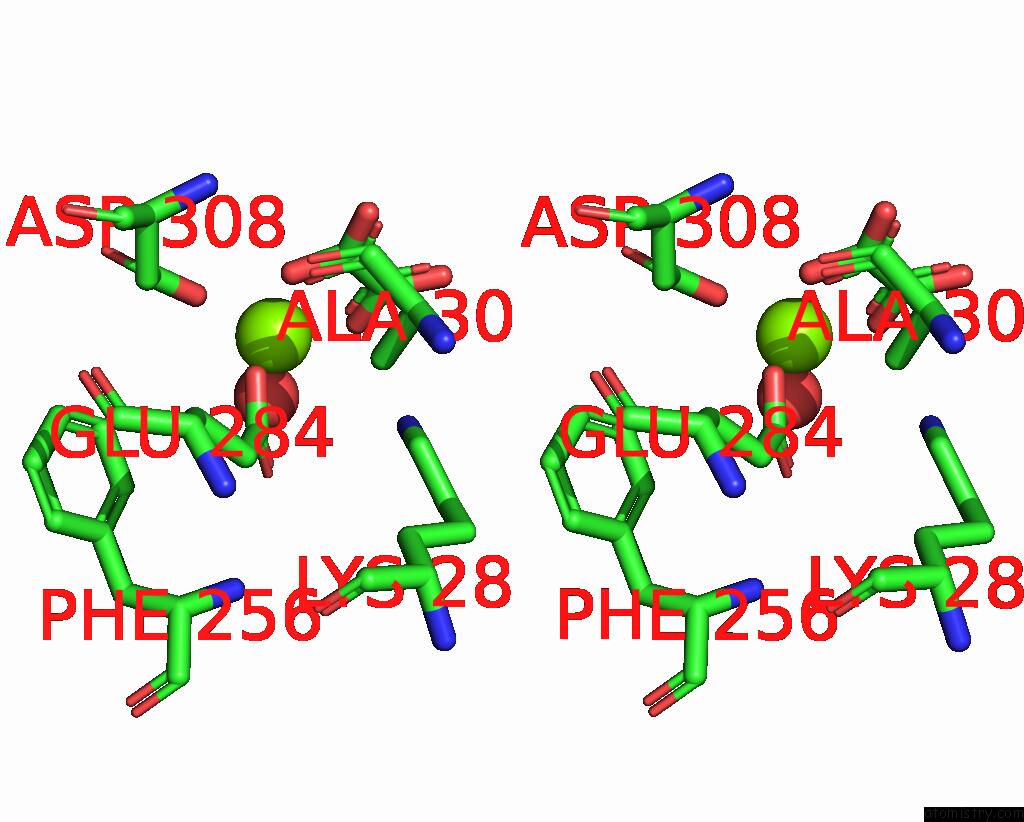
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 7 of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58 within 5.0Å range:
|
Magnesium binding site 8 out of 8 in 5scd
Go back to
Magnesium binding site 8 out
of 8 in the Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58
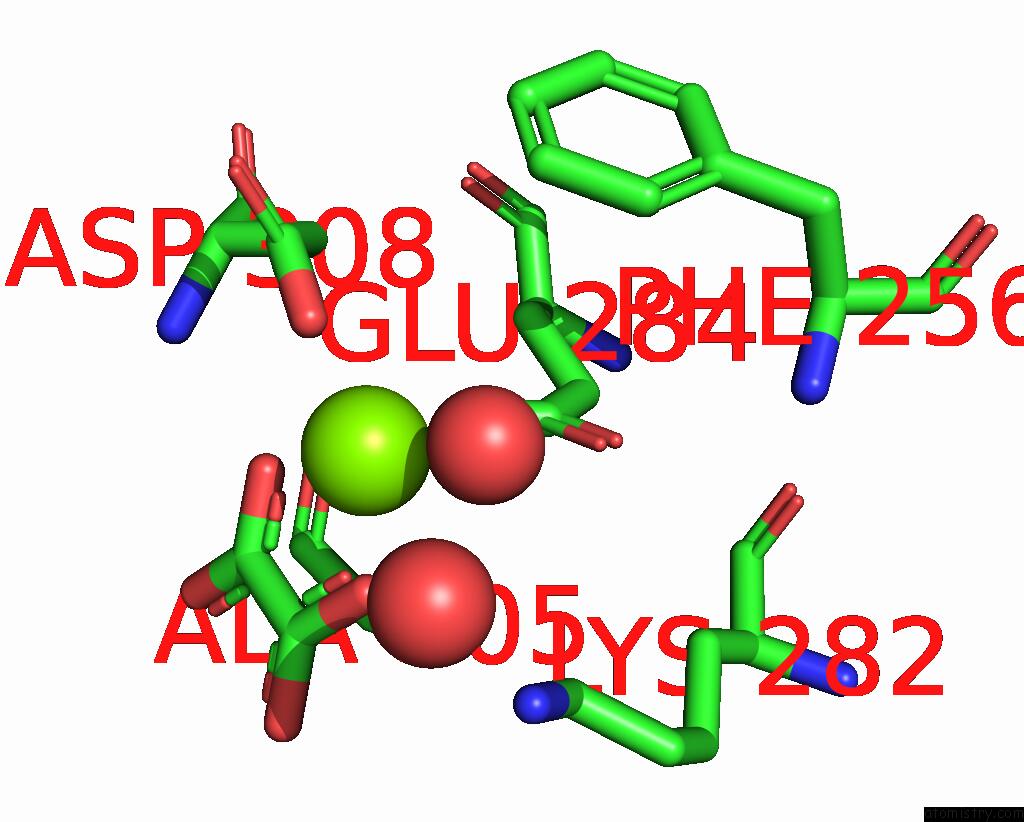
Mono view
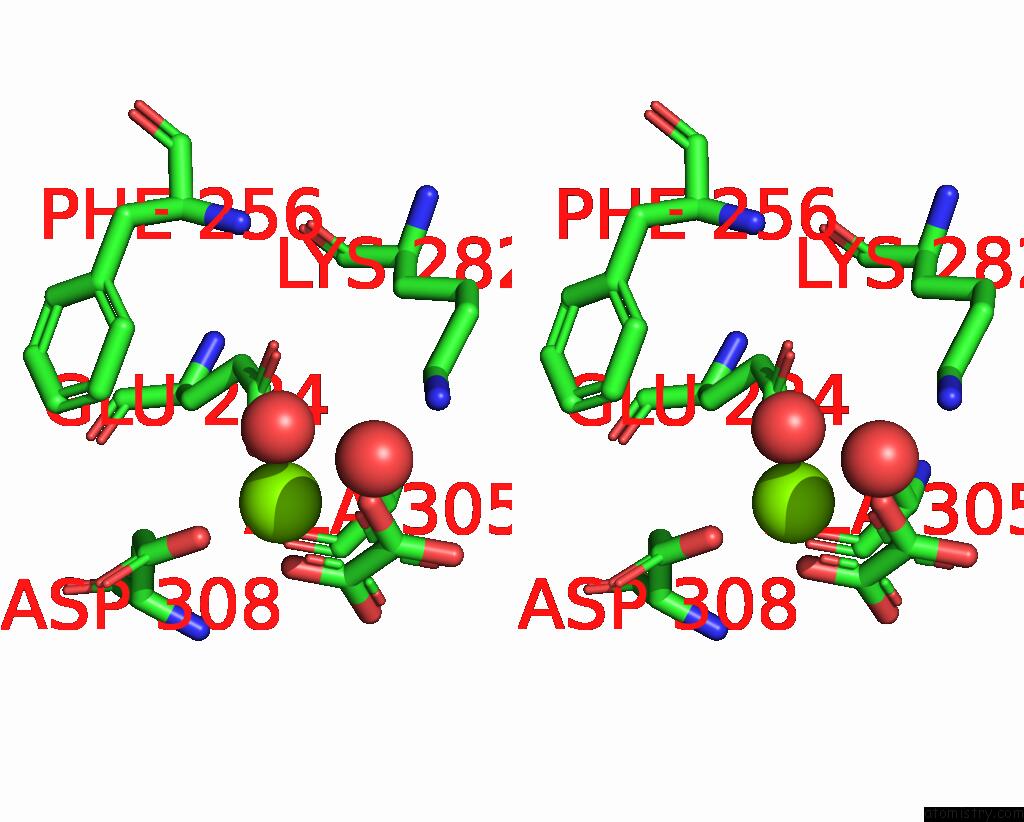
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 8 of Structure of Liver Pyruvate Kinase in Complex with Anthraquinone Derivative 58 within 5.0Å range:
|
Reference:
A.Nain-Perez,
A.Foller Fuchtbauer,
L.Haversen,
A.Lulla,
C.Gao,
J.Matic,
L.Monjas,
A.Rodriguez,
P.Brear,
W.Kim,
M.Hyvonen,
J.Boren,
A.Mardinoglu,
M.Uhlen,
M.Grotli.
Anthraquinone Derivatives As Adp-Competitive Inhibitors of Liver Pyruvate Kinase. Eur.J.Med.Chem. V. 234 14270 2022.
ISSN: ISSN 0223-5234
PubMed: 35290845
DOI: 10.1016/J.EJMECH.2022.114270
Page generated: Mon Sep 30 03:06:43 2024
ISSN: ISSN 0223-5234
PubMed: 35290845
DOI: 10.1016/J.EJMECH.2022.114270
Last articles
Zn in 9J0NZn in 9J0O
Zn in 9J0P
Zn in 9FJX
Zn in 9EKB
Zn in 9C0F
Zn in 9CAH
Zn in 9CH0
Zn in 9CH3
Zn in 9CH1