Magnesium »
PDB 7nem-7noz »
7njl »
Magnesium in PDB 7njl: Mycobacterium Smegmatis Atp Synthase State 1B
Enzymatic activity of Mycobacterium Smegmatis Atp Synthase State 1B
All present enzymatic activity of Mycobacterium Smegmatis Atp Synthase State 1B:
7.1.2.2;
7.1.2.2;
Magnesium Binding Sites:
The binding sites of Magnesium atom in the Mycobacterium Smegmatis Atp Synthase State 1B
(pdb code 7njl). This binding sites where shown within
5.0 Angstroms radius around Magnesium atom.
In total 5 binding sites of Magnesium where determined in the Mycobacterium Smegmatis Atp Synthase State 1B, PDB code: 7njl:
Jump to Magnesium binding site number: 1; 2; 3; 4; 5;
In total 5 binding sites of Magnesium where determined in the Mycobacterium Smegmatis Atp Synthase State 1B, PDB code: 7njl:
Jump to Magnesium binding site number: 1; 2; 3; 4; 5;
Magnesium binding site 1 out of 5 in 7njl
Go back to
Magnesium binding site 1 out
of 5 in the Mycobacterium Smegmatis Atp Synthase State 1B
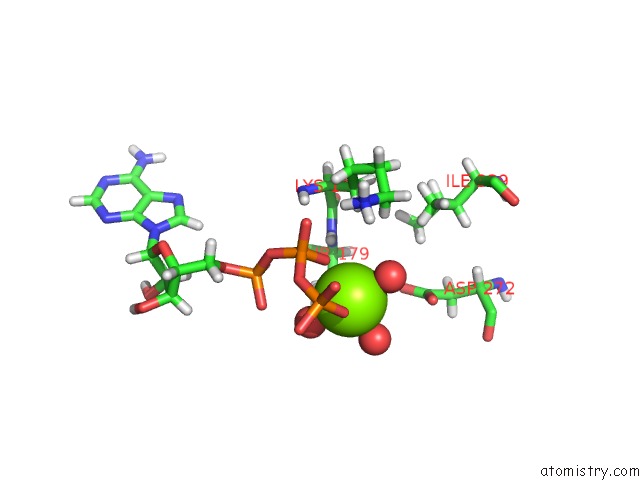
Mono view
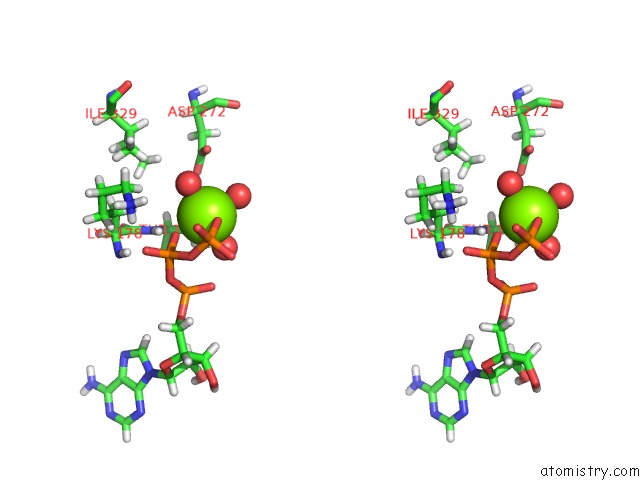
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 1 of Mycobacterium Smegmatis Atp Synthase State 1B within 5.0Å range:
|
Magnesium binding site 2 out of 5 in 7njl
Go back to
Magnesium binding site 2 out
of 5 in the Mycobacterium Smegmatis Atp Synthase State 1B
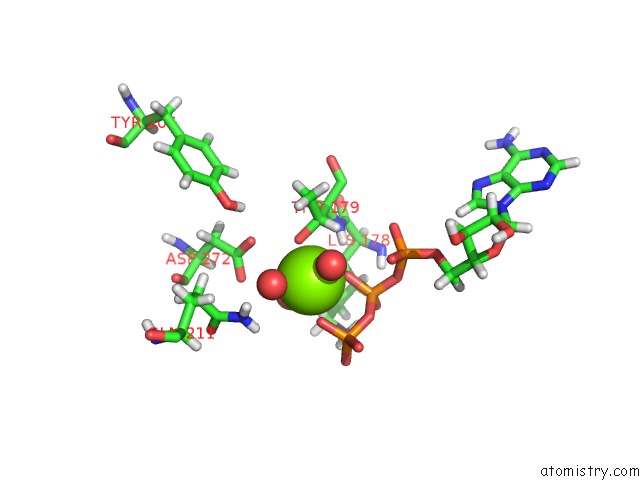
Mono view
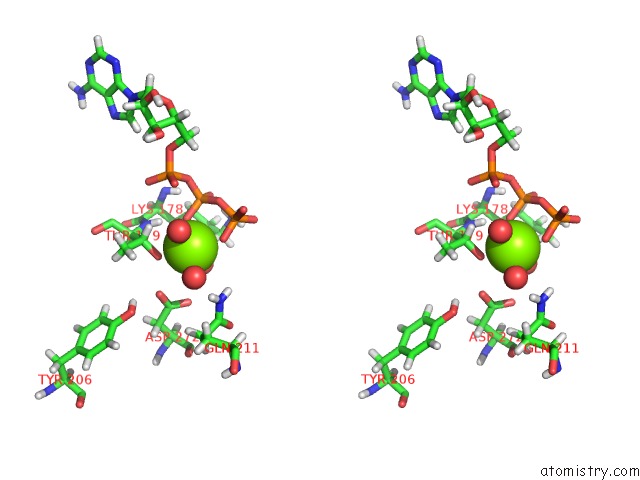
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 2 of Mycobacterium Smegmatis Atp Synthase State 1B within 5.0Å range:
|
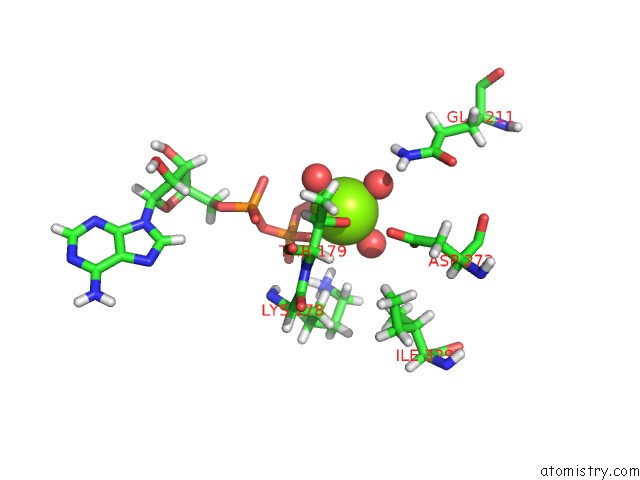
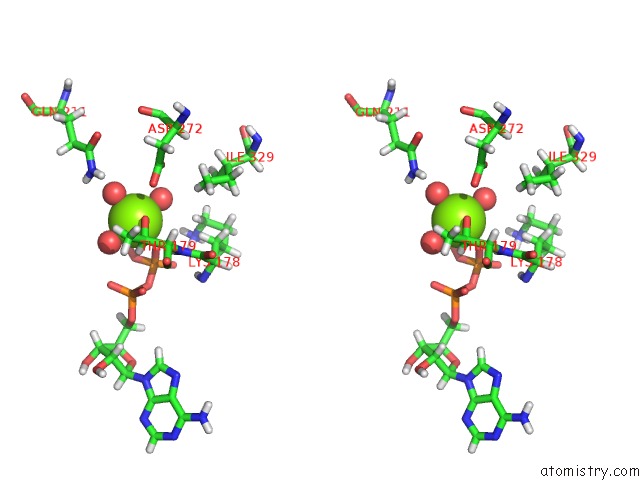
Magnesium binding site 3 out of 5 in 7njl
Go back to
Magnesium binding site 3 out
of 5 in the Mycobacterium Smegmatis Atp Synthase State 1B
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 3 of Mycobacterium Smegmatis Atp Synthase State 1B within 5.0Å range:
|
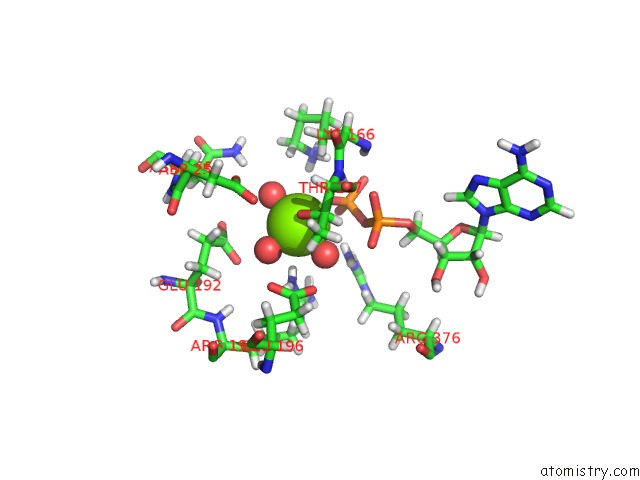
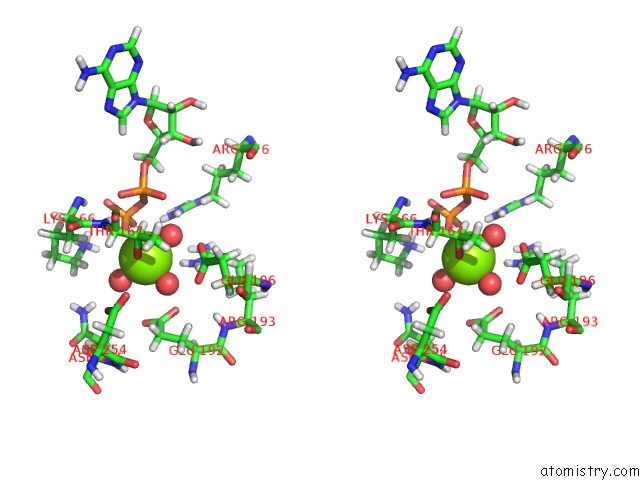
Magnesium binding site 4 out of 5 in 7njl
Go back to
Magnesium binding site 4 out
of 5 in the Mycobacterium Smegmatis Atp Synthase State 1B
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 4 of Mycobacterium Smegmatis Atp Synthase State 1B within 5.0Å range:
|
Magnesium binding site 5 out of 5 in 7njl
Go back to
Magnesium binding site 5 out
of 5 in the Mycobacterium Smegmatis Atp Synthase State 1B
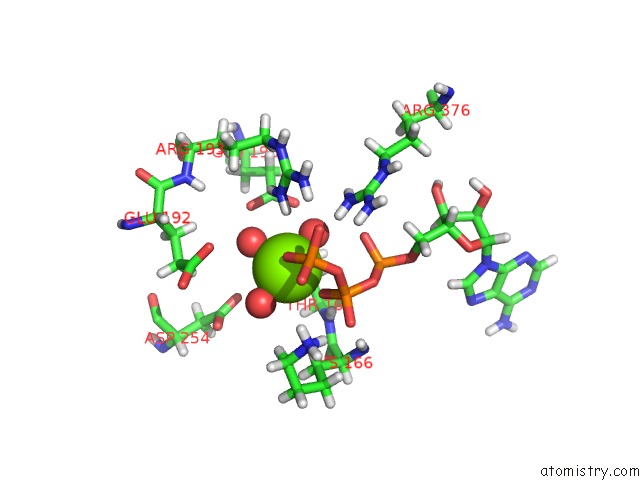
Mono view
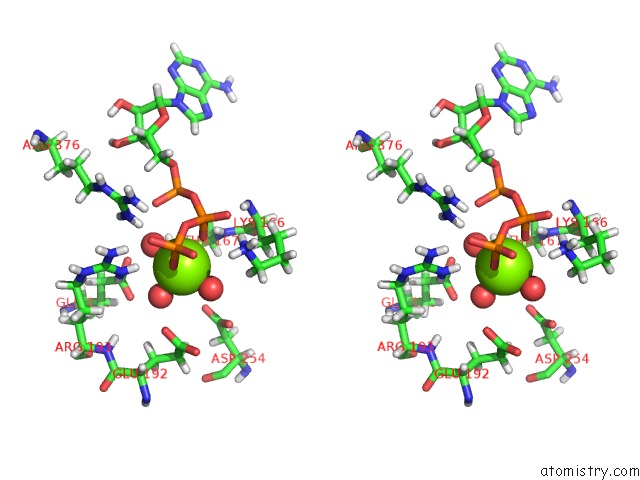
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 5 of Mycobacterium Smegmatis Atp Synthase State 1B within 5.0Å range:
|
Reference:
J.Petri,
M.G.Montgomery,
T.J.Spikes,
G.M.Cook,
J.E.Walker.
Structure of the Atp Synthase From Mycobacterium Smegmatis To Be Published.
Page generated: Thu Oct 3 02:05:15 2024
Last articles
Zn in 9MJ5Zn in 9HNW
Zn in 9G0L
Zn in 9FNE
Zn in 9DZN
Zn in 9E0I
Zn in 9D32
Zn in 9DAK
Zn in 8ZXC
Zn in 8ZUF