Magnesium »
PDB 1bho-1c5p »
1bzy »
Magnesium in PDB 1bzy: Human Hgprtase with Transition State Inhibitor
Enzymatic activity of Human Hgprtase with Transition State Inhibitor
All present enzymatic activity of Human Hgprtase with Transition State Inhibitor:
2.4.2.8;
2.4.2.8;
Protein crystallography data
The structure of Human Hgprtase with Transition State Inhibitor, PDB code: 1bzy
was solved by
W.Shi,
C.Li,
P.C.Tyler,
R.H.Furneaux,
C.Grubmeyer,
V.L.Schramm,
S.C.Almo,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 10.00 / 2.00 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 52.960, 102.010, 144.270, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 19 / 24.9 |
Magnesium Binding Sites:
The binding sites of Magnesium atom in the Human Hgprtase with Transition State Inhibitor
(pdb code 1bzy). This binding sites where shown within
5.0 Angstroms radius around Magnesium atom.
In total 8 binding sites of Magnesium where determined in the Human Hgprtase with Transition State Inhibitor, PDB code: 1bzy:
Jump to Magnesium binding site number: 1; 2; 3; 4; 5; 6; 7; 8;
In total 8 binding sites of Magnesium where determined in the Human Hgprtase with Transition State Inhibitor, PDB code: 1bzy:
Jump to Magnesium binding site number: 1; 2; 3; 4; 5; 6; 7; 8;
Magnesium binding site 1 out of 8 in 1bzy
Go back to
Magnesium binding site 1 out
of 8 in the Human Hgprtase with Transition State Inhibitor
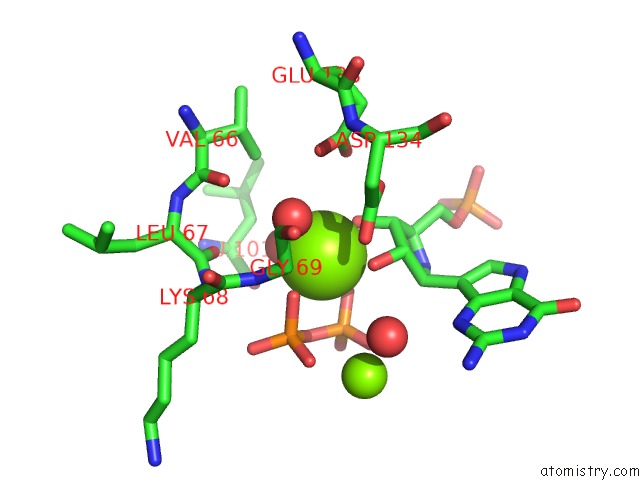
Mono view
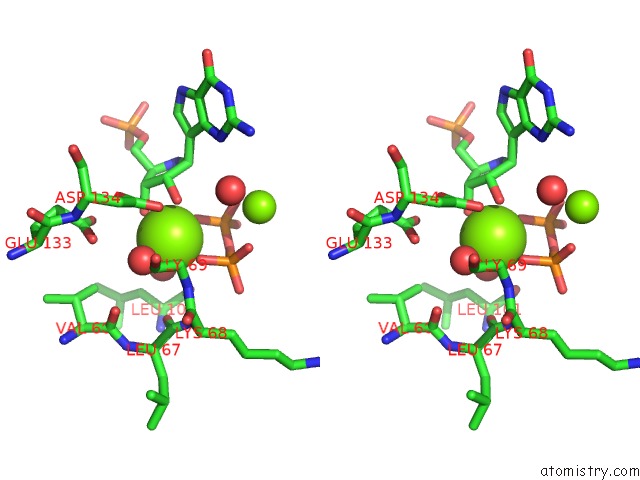
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 1 of Human Hgprtase with Transition State Inhibitor within 5.0Å range:
|
Magnesium binding site 2 out of 8 in 1bzy
Go back to
Magnesium binding site 2 out
of 8 in the Human Hgprtase with Transition State Inhibitor
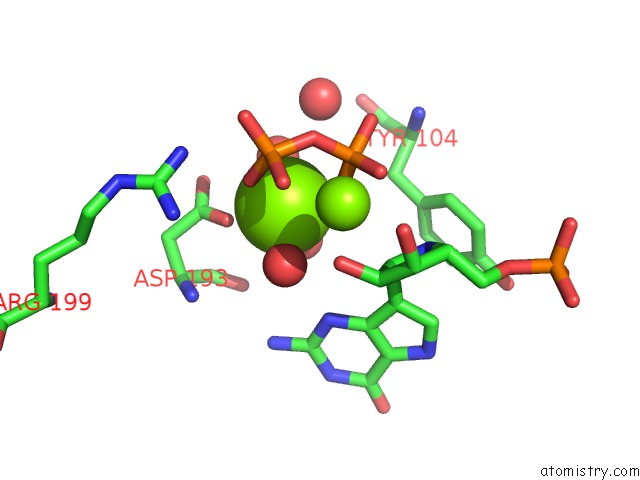
Mono view
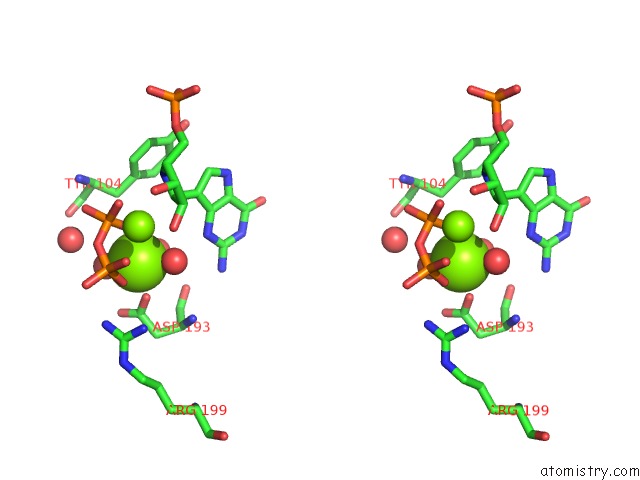
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 2 of Human Hgprtase with Transition State Inhibitor within 5.0Å range:
|
Magnesium binding site 3 out of 8 in 1bzy
Go back to
Magnesium binding site 3 out
of 8 in the Human Hgprtase with Transition State Inhibitor
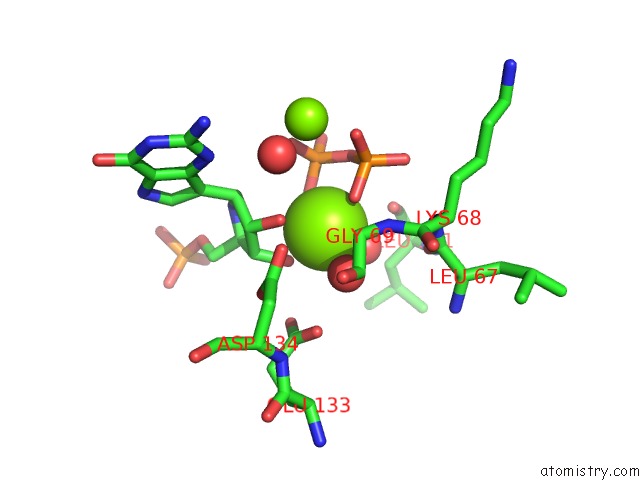
Mono view
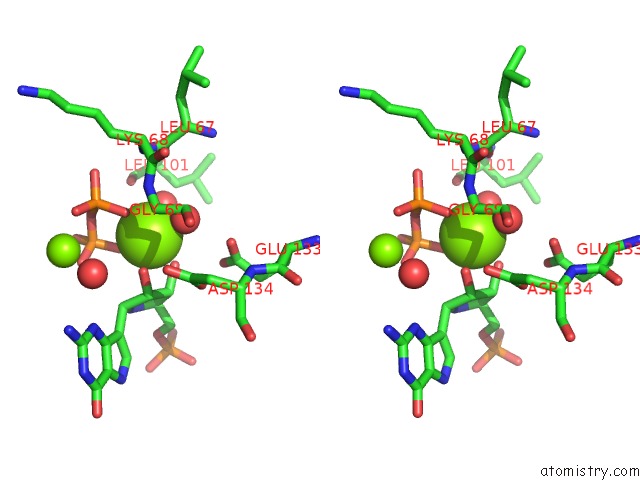
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 3 of Human Hgprtase with Transition State Inhibitor within 5.0Å range:
|
Magnesium binding site 4 out of 8 in 1bzy
Go back to
Magnesium binding site 4 out
of 8 in the Human Hgprtase with Transition State Inhibitor
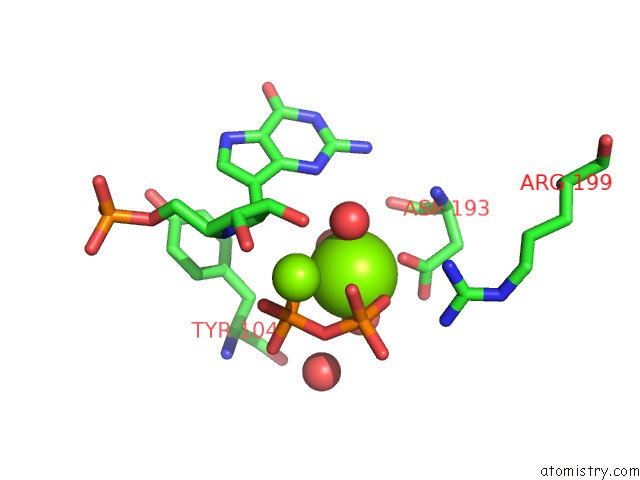
Mono view
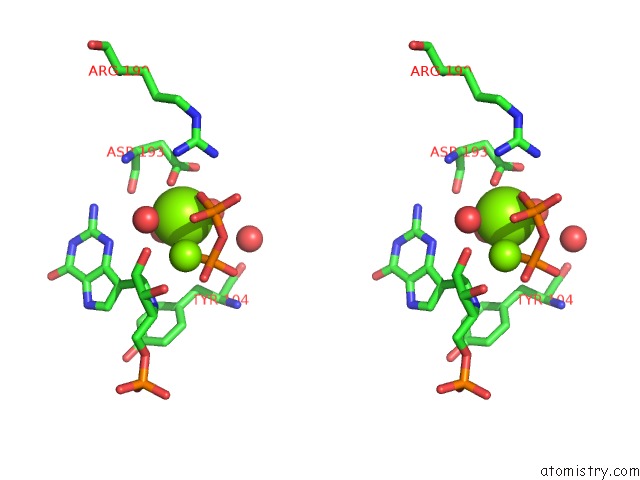
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 4 of Human Hgprtase with Transition State Inhibitor within 5.0Å range:
|
Magnesium binding site 5 out of 8 in 1bzy
Go back to
Magnesium binding site 5 out
of 8 in the Human Hgprtase with Transition State Inhibitor
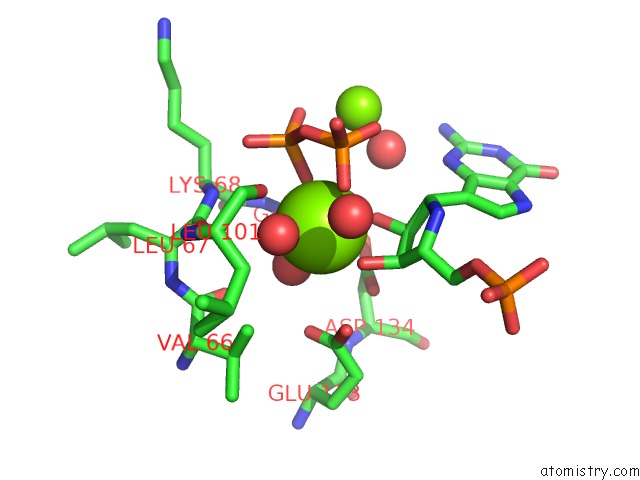
Mono view
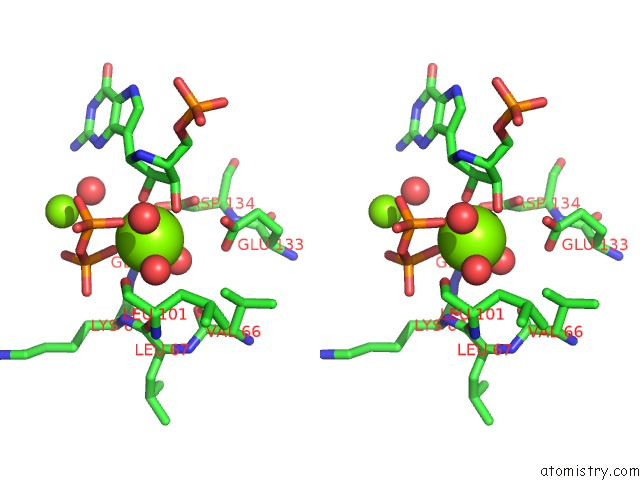
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 5 of Human Hgprtase with Transition State Inhibitor within 5.0Å range:
|
Magnesium binding site 6 out of 8 in 1bzy
Go back to
Magnesium binding site 6 out
of 8 in the Human Hgprtase with Transition State Inhibitor
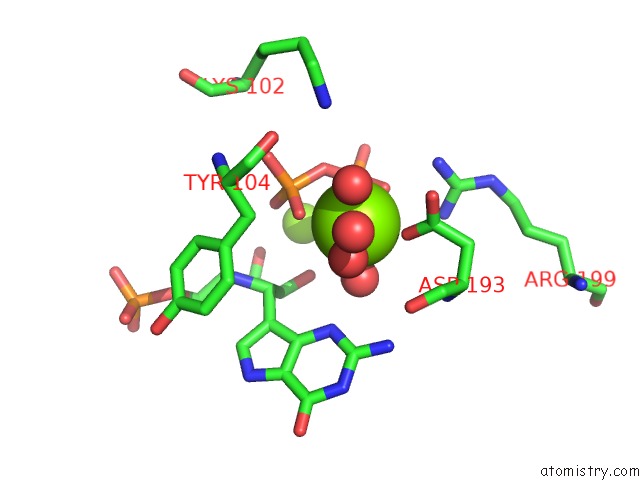
Mono view
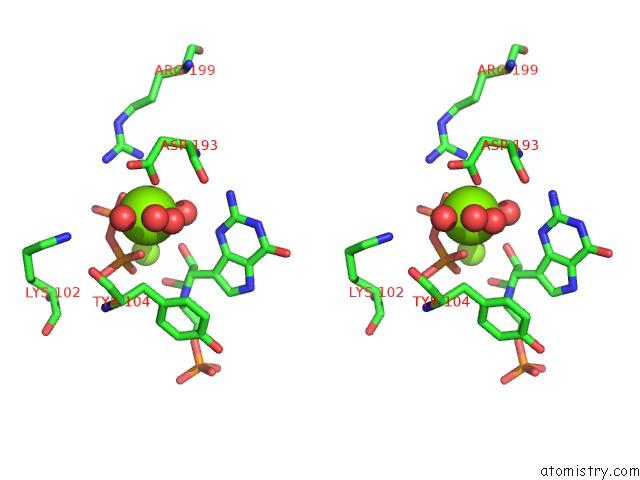
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 6 of Human Hgprtase with Transition State Inhibitor within 5.0Å range:
|
Magnesium binding site 7 out of 8 in 1bzy
Go back to
Magnesium binding site 7 out
of 8 in the Human Hgprtase with Transition State Inhibitor
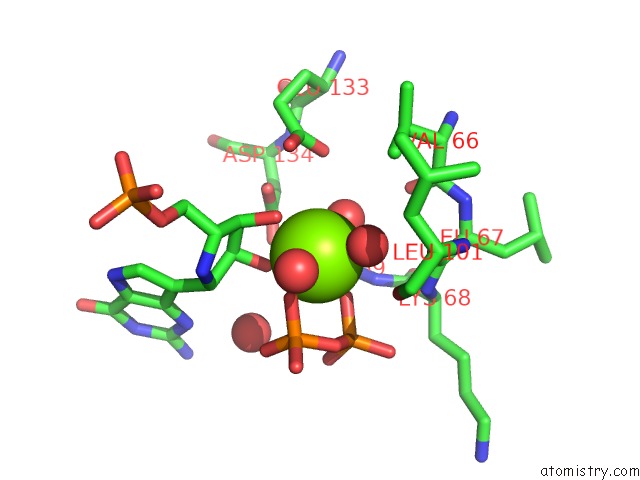
Mono view
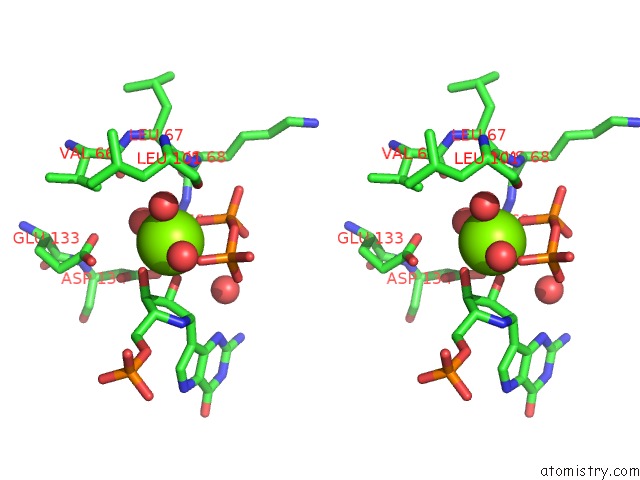
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 7 of Human Hgprtase with Transition State Inhibitor within 5.0Å range:
|
Magnesium binding site 8 out of 8 in 1bzy
Go back to
Magnesium binding site 8 out
of 8 in the Human Hgprtase with Transition State Inhibitor
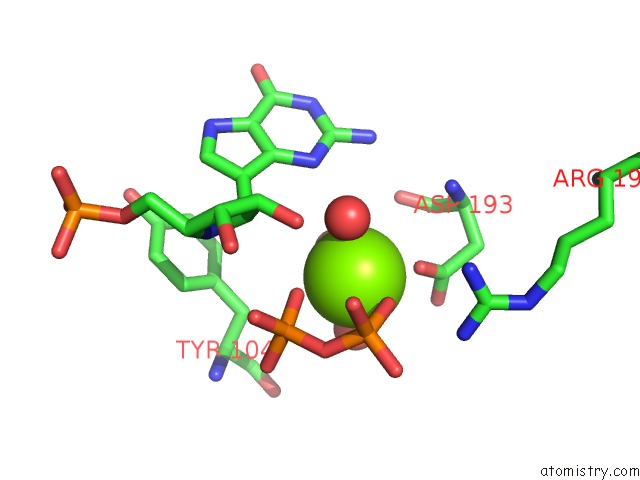
Mono view
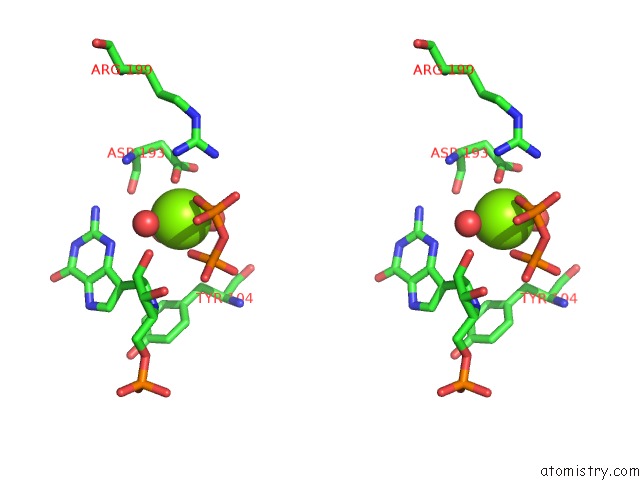
Stereo pair view

Mono view

Stereo pair view
A full contact list of Magnesium with other atoms in the Mg binding
site number 8 of Human Hgprtase with Transition State Inhibitor within 5.0Å range:
|
Reference:
W.Shi,
C.M.Li,
P.C.Tyler,
R.H.Furneaux,
C.Grubmeyer,
V.L.Schramm,
S.C.Almo.
The 2.0 A Structure of Human Hypoxanthine-Guanine Phosphoribosyltransferase in Complex with A Transition-State Analog Inhibitor. Nat.Struct.Biol. V. 6 588 1999.
ISSN: ISSN 1072-8368
PubMed: 10360366
DOI: 10.1038/9376
Page generated: Sat Aug 9 20:14:47 2025
ISSN: ISSN 1072-8368
PubMed: 10360366
DOI: 10.1038/9376
Last articles
Mg in 1SL2Mg in 1SL5
Mg in 1SKR
Mg in 1SL0
Mg in 1SL1
Mg in 1SKW
Mg in 1SKS
Mg in 1SJC
Mg in 1SJB
Mg in 1SKQ